47627
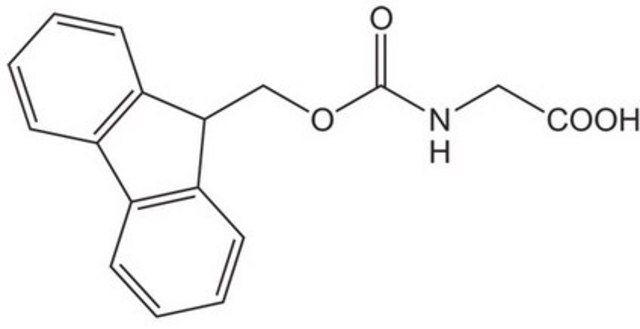
Fmoc-Gly-OH
≥98.0% (T)
Synonyme(s) :
Fmoc-glycine
About This Item
Produits recommandés
Niveau de qualité
Pureté
≥98.0% (T)
Forme
powder
Capacité de réaction
reaction type: Fmoc solid-phase peptide synthesis
Pf
174-175 °C (lit.)
174-178 °C
Application(s)
peptide synthesis
Groupe fonctionnel
Fmoc
Température de stockage
2-8°C
Chaîne SMILES
OC(=O)CNC(=O)OCC1c2ccccc2-c3ccccc13
InChI
1S/C17H15NO4/c19-16(20)9-18-17(21)22-10-15-13-7-3-1-5-11(13)12-6-2-4-8-14(12)15/h1-8,15H,9-10H2,(H,18,21)(H,19,20)
Clé InChI
NDKDFTQNXLHCGO-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Application
- Efficient Fmoc-Protected Amino Ester Hydrolysis Using Green Calcium (II) Iodide as a Protective Agent: This study describes the utilization of environmentally friendly calcium iodide in the hydrolysis of Fmoc-protected amino esters, enhancing reaction efficiency and sustainability (R Binette, M Desgagné, C Theaud, PL Boudreault - Molecules, 2022). Link to the article.
- α/β-Chimera peptide synthesis with cyclic β-sugar amino acids: the efficient coupling protocol: This research provides an advanced synthesis method for α/β-chimera peptides using cyclic β-sugar amino acids, demonstrating significant implications for peptide design in medicinal chemistry (A Nagy, V Goldschmidt Gőz, I Pintér, V Farkas - Amino acids, 2019). Link to the article.
- MS, CD, and FT-IR characterization of five newly synthesized histidine-containing Ala-and Gly-based peptides: This paper presents detailed characterization of novel histidine-containing peptides, highlighting techniques that could be pivotal for peptide-based drug discovery (M Murariu, L Ion, CI Ciobanu, BA Petre - Rev. Roum Chem., 2017). Link to the article.
- Efficient method for the concentration determination of fmoc groups incorporated in the core-shell materials by Fmoc–glycine: This article elaborates on an efficient method for determining the concentration of fmoc groups in core-shell materials, critical for the design of advanced functional materials (E Szczepańska, B Grobelna, J Ryl, A Kulpa - Molecules, 2020). Link to the article.
- Circular aqueous fmoc/t‐bu solid‐phase peptide synthesis: This study explores a novel approach in solid-phase peptide synthesis, utilizing circular aqueous techniques that may offer greener and more efficient methodologies for peptide synthesis (J Pawlas, JH Rasmussen - ChemSusChem, 2021). Link to the article.
Autres remarques
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
With a growing peptide drug market the fast, reliable and uncomplicated synthesis of peptides is of paramount importance.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique