456055
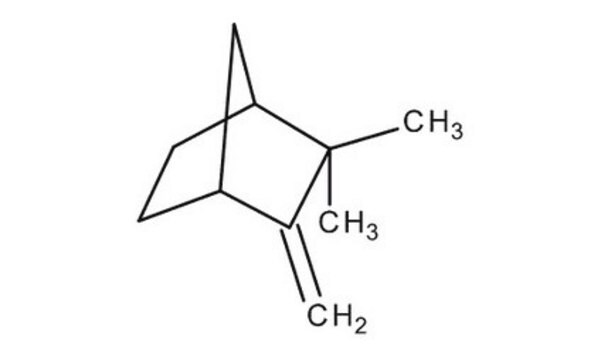
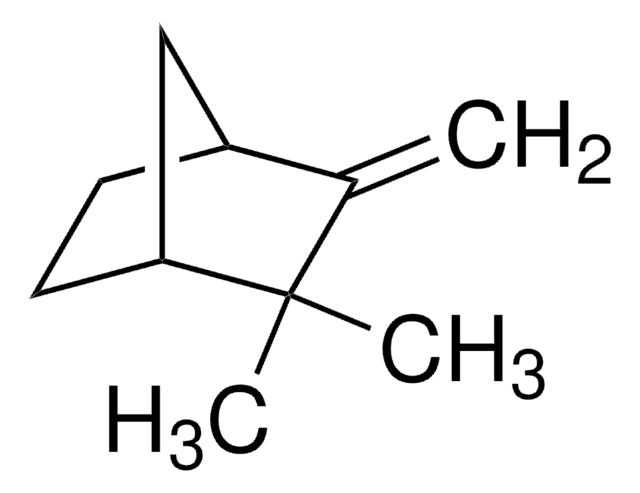
Camphene
95%
Synonyme(s) :
(±)-Camphene, 2,2-Dimethyl-3-methylenebicyclo[2.2.1]heptane, 2,2-Dimethyl-3-methylenenorbornane, 2,2-Dimethyl-3-methylidenebicyclo[2.2.1]heptane, 2-Methylene-3,3-dimethylbicyclo[2.2.1]heptane, 3,3-Dimethyl-2-methylenenorbornane, 3,3-Dimethyl-2-methylenenorcamphane, DL-Camphene
About This Item
Produits recommandés
Niveau de qualité
Essai
95%
Forme
solid
pb
159-160 °C (lit.)
Pf
48-52 °C (lit.)
Densité
0.85 g/mL at 25 °C (lit.)
Chaîne SMILES
[H][C@]12CC[C@]([H])(C1)C(C)(C)C2=C
InChI
1S/C10H16/c1-7-8-4-5-9(6-8)10(7,2)3/h8-9H,1,4-6H2,2-3H3/t8-,9+/m0/s1
Clé InChI
CRPUJAZIXJMDBK-DTWKUNHWSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- Isobornyl carboxylates by silica-supported tungstophosphoric acid-catalyzed liquid-phase esterification of C2-C6 fatty acids.
- Hydroaminated camphene via intermolecular anti-Markovnikov hydroamination reaction with N-hydroxyphthalimide and triethyl phosphite in the presence of dilauroyl peroxide as an initiator.
- Camphene oxide via methyltrioxorhenium-catalyzed epoxidation in the presence of H2O2 as an oxidant and pyrazole as a Lewis base adduct.
- Isobornyl alkyl ethers using alcohols via cation exchange resin-catalyzed alkoxylation.
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Flam. Sol. 1
Code de la classe de stockage
4.1B - Flammable solid hazardous materials
Classe de danger pour l'eau (WGK)
WGK 2
Point d'éclair (°F)
78.8 °F - DIN 51755 Part 1
Point d'éclair (°C)
26 °C - DIN 51755 Part 1
Équipement de protection individuelle
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Protocoles
-Pinocarveol; Menthol; (+)-Terpinen-4-ol; α-Terpineol; (±)-α-Terpinyl acetate, predominantly α-isomer; Germacrene D
Cymene; 4,5,6,7-Tetrahydro-3,6-dimethylbenzofuran; Linalool; Menthol; Menthone; Menthyl acetate; Germacrene D; Bicyclogermacrene; Thymol
-α-Bergamotene; β-Bisabolene; α-Terpineol; Neryl acetate; Geranyl acetate; Neral; Geranial
-3,7-Dimethyl-2,6-octadien-1-ol; Neral; Geraniol; Geranial; Undecanal; Citronellyl acetate; Neryl acetate; 3,7-Dimethyl-2,6-octadienyl acetate; 1-Tetradecene; Tetradecane; α-Bisabolol
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique