345474
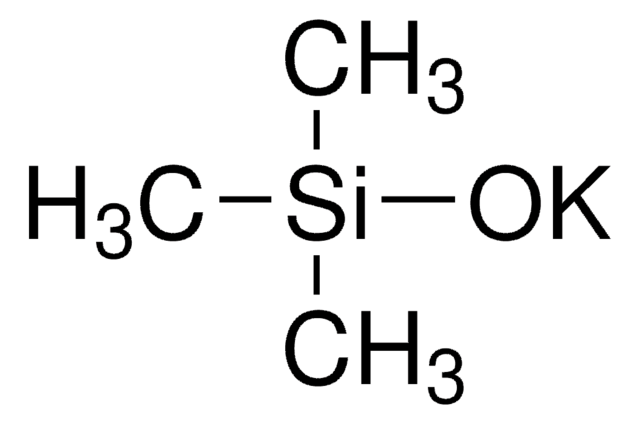
Lithium trimethylsilanolate
95%
Synonyme(s) :
Trimethylsilanol lithium salt
About This Item
Produits recommandés
Niveau de qualité
Pureté
95%
Forme
solid
Chaîne SMILES
[Li+].C[Si](C)(C)[O-]
InChI
1S/C3H9OSi.Li/c1-5(2,3)4;/h1-3H3;/q-1;+1
Clé InChI
OXOZHAWWRPCVGL-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Application
- Enhanced process and composition control for atomic layer deposition with lithium trimethylsilanolate: This study reports on the novel lithium precursor, lithium trimethylsilanolate (LiTMSO), for atomic layer deposition, comparing it with other lithium precursors for improved process and composition control (Ruud et al., 2017).
- Trimethylsiloxy based metal complexes as electrolyte additives for high voltage application in lithium ion cells: The paper discusses the synthesis of lithium trimethylsilanolate and its use as an additive to enhance the performance of high voltage lithium ion cells (Imholt et al., 2017).
- Application of trimethylsilanolate alkali salts in organic synthesis: This review covers various applications of lithium trimethylsilanolate in organic synthesis, highlighting its role in different reactions (Bürglová and Hlaváč, 2018).
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Eye Dam. 1 - Skin Corr. 1B
Code de la classe de stockage
8A - Combustible corrosive hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Équipement de protection individuelle
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique