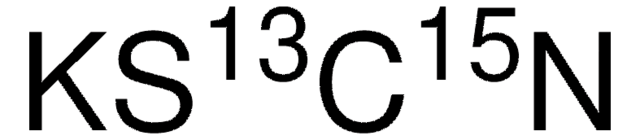167134
Bromohydroquinone
97%
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule linéaire :
BrC6H3(OH)2
Numéro CAS:
Poids moléculaire :
189.01
Numéro CE :
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Essai
97%
Pf
112-116 °C (lit.)
Groupe fonctionnel
bromo
Chaîne SMILES
Oc1ccc(O)c(Br)c1
InChI
1S/C6H5BrO2/c7-5-3-4(8)1-2-6(5)9/h1-3,8-9H
Clé InChI
REFDOIWRJDGBHY-UHFFFAOYSA-N
Catégories apparentées
Application
Bromohydroquinone was used in the synthesis of Π-conjugated polymers composed of alkyl carbazole/dialkoxyphenylene and squaraine units via Sonogashira cross-coupling reactions. It was used in the preparation of 2-bromobenzoquinone.
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
T J Monks et al.
Molecular pharmacology, 34(1), 15-22 (1988-07-01)
The formation of potentially reactive thiols has been postulated to play a role in the nephrotoxicity caused by a number of glutathione and/or cysteine conjugates. However, the inherent reactivity of such compounds has precluded both their identification in biological systems
T J Monks et al.
Drug metabolism and disposition: the biological fate of chemicals, 13(5), 553-559 (1985-09-01)
Incubation of either o-bromophenol or 2-bromohydroquinone with rat liver microsomes and 0.25 mM 35S-glutathione (GSH) gave rise to several isomeric 35S-GSH conjugates. A mixture of these isomeric GSH conjugates was prepared chemically and two were purified by HPLC; 1H-NMR spectroscopy
R G Schnellmann
Toxicology and applied pharmacology, 99(1), 11-18 (1989-06-01)
2-Bromohydroquinone (BHQ) plays an important role in bromobenzene-induced nephrotoxicity and is a model toxic hydroquinone. Since BHQ has a quinone nucleus and various quinones have been shown to produce cytotoxicity via oxidative stress, the goal of this study was to
G W Miller et al.
Toxicology and applied pharmacology, 125(2), 192-197 (1994-04-01)
We have previously shown that strychnine mimics the cytoprotective properties of glycine in renal proximal tubule (RPT) suspensions exposed to antimycin A (AA). The aims of this study were to determine whether the cytoprotective properties of strychnine applied to various
S S Lau et al.
Biochemical and biophysical research communications, 152(1), 223-230 (1988-04-15)
2-Bromo-(diglutathion-Syl)hydroquinone (2-Br-[diGSyl]HQ) is a more potent nephrotoxicant than any of three mono-substituted isomers. The reason for this differential toxicity is unknown. We now report that the rate of uptake of 2-Br-(diGSyl)HQ, 2-Br-3-(GSyl)HQ, 2-Br-5-(GSyl)-HQ and 2-Br-6(GSyl)HQ by kidney slices was 2.4
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique