H17082
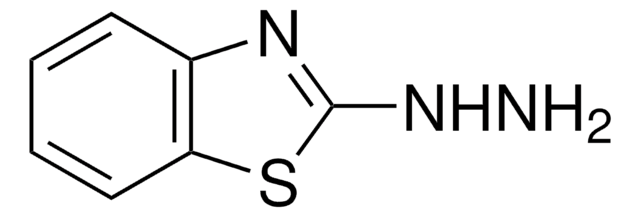
2-Hydrazinopyridine
97%
Synonym(s):
2-Pyridylhydrazine
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C5H7N3
CAS Number:
Molecular Weight:
109.13
Beilstein:
109984
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
bp
90-92 °C/1 mmHg (lit.)
mp
41-44 °C (lit.)
storage temp.
2-8°C
SMILES string
NNc1ccccn1
InChI
1S/C5H7N3/c6-8-5-3-1-2-4-7-5/h1-4H,6H2,(H,7,8)
InChI key
NWELCUKYUCBVKK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Enhanced Metal Removal: Utilizing Schiff base functionalized dialdehyde starch, derived from 2-hydrazinopyridine, demonstrated significant potential in enhancing the removal of Cu(II) from solutions. The study included preparation methodologies, performance evaluations, and DFT calculations, showcasing its efficacy in water treatment technologies (Liang et al., 2024).
- Dual Sensing Probe Development: A novel dicyanisophorone-based probe, incorporating 2-hydrazinopyridine, was developed for the dual sensing of Zn(2+) and Cd(2+) via near-infrared fluorescence. This advancement aids in the detection and analysis of heavy metals in various environmental and biological samples (Yan et al., 2023).
- Active Site Analysis in Lysyl Oxidase: The study provided insights into the spatial arrangement of active site components in Lysyl Oxidase-like 2, including the role of 2-hydrazinopyridine, which is critical for understanding the enzyme′s mechanism and potential therapeutic applications (Meier et al., 2022).
- Structural Analysis of Lysyl Oxidase: Research focused on the predicted 3D structure of the amine oxidase domain of Lysyl Oxidase-Like 2, exploring the interaction dynamics facilitated by 2-hydrazinopyridine. This contributes significantly to the field of molecular biology and enzyme function analysis (Meier et al., 2022).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
E S H El Ashry et al.
Nucleosides, nucleotides & nucleic acids, 23(3), 567-580 (2004-04-29)
Reaction of 2-hydrazinopyridine (1) with D-xylose, D-galactose, D-glucose and D-fructose afforded the corresponding hydrazones mainly in the acyclic forms 2, 3, 6 and 11 with minor amounts of the cyclic structures. Oxidative cyclization of the hydrazones with bromine in methanol
C M Wilmot et al.
Biochemistry, 36(7), 1608-1620 (1997-02-18)
The crystal structure of the complex between the copper amine oxidase from Escherichia coli (ECAO) and a covalently bound inhibitor, 2-hydrazinopyridine, has been determined to a resolution of 2.0 A. The inhibitor covalently binds at the 5 position of the
G De Matteis et al.
Journal of biological inorganic chemistry : JBIC : a publication of the Society of Biological Inorganic Chemistry, 4(3), 348-353 (1999-08-10)
Bovine serum amine oxidase (BSAO) reacts with 2-hydrazinopyridine, which binds the organic co-factor 2,4,5-trihydroxyphenylalanine quinone, forming a band at 435 nm. The band shifts to 526 nm around 60 degrees C, to 415 nm upon denaturation, but only shifts to
Conserved tyrosine-369 in the active site of Escherichia coli copper amine oxidase is not essential.
J M Murray et al.
Biochemistry, 40(43), 12808-12818 (2001-10-24)
Copper amine oxidases are homodimeric enzymes that catalyze two reactions: first, a self-processing reaction to generate the 2,4,5-trihydroxyphenylalanine (TPQ) cofactor from an active site tyrosine by a single turnover mechanism; second, the oxidative deamination of primary amine substrates with the
Minae Mure et al.
Biochemistry, 44(5), 1583-1594 (2005-02-03)
Adduct I (lambda(max) at approximately 430 nm) formed in the reaction of 2-hydrazinopyridine (2HP) and the TPQ cofactor of wild-type Escherichia coli copper amine oxidase (WT-ECAO) is stable at neutral pH, 25 degrees C, but slowly converts to another spectroscopically
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service