917974
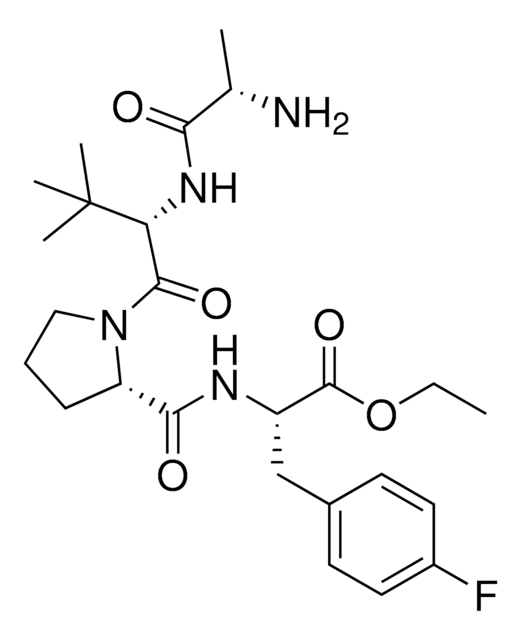
BocA1V2PF2
Synonym(s):
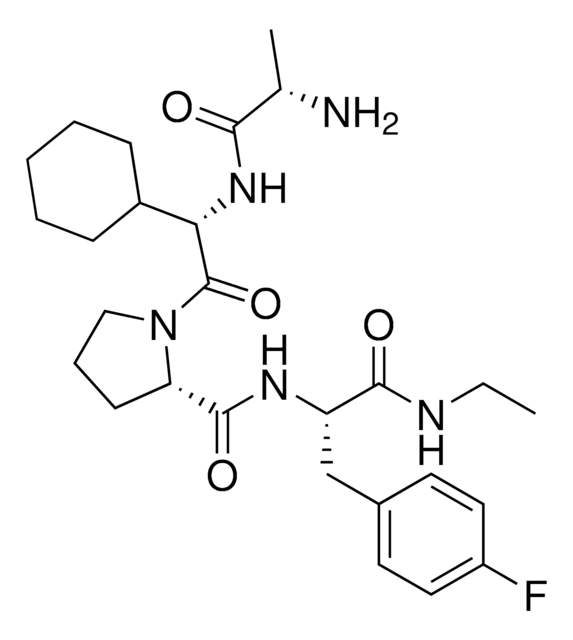
(S)-2-((S)-1-((S)-2-((S)-2-((tert-Butoxycarbonyl)amino)propanamido)-2-cyclohexylacetyl)pyrrolidine-2-carboxamido)-3-(4-fluorophenyl)propanoic acid, IAP E3 ligase lead for protein degrader research, SNIPER building block
About This Item
Recommended Products
ligand
BocA1V2PF2
Quality Level
form
powder
reaction suitability
reagent type: ligand
functional group
carboxylic acid
storage temp.
2-8°C
SMILES string
C[C@H](NC(OC(C)(C)C)=O)C(N[C@H](C(N1CCC[C@H]1C(N[C@H](C(O)=O)CC2=CC=C(C=C2)F)=O)=O)C3CCCCC3)=O
Application
BocA1V2PF2 conjugates are also available for degrader synthesis. Browse our full synthesis offering here for streamlining
SNIPER and PROTAC® degrader libraries: Degrader Building Blocks
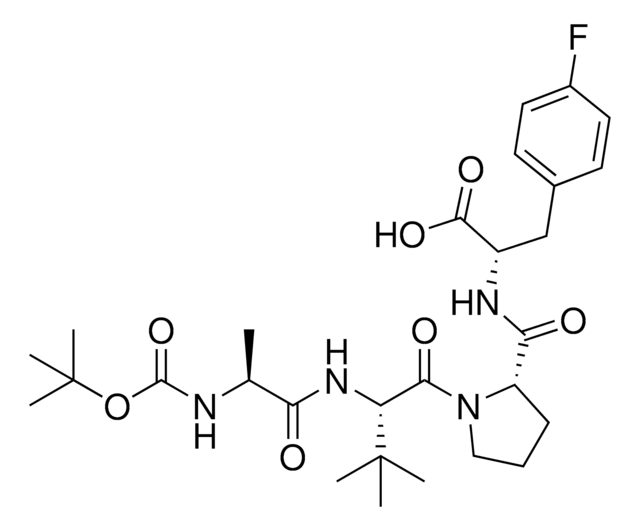
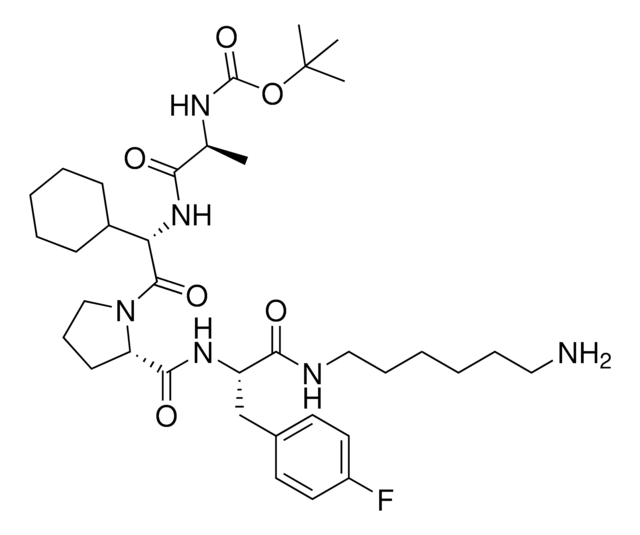
917443BocA1V2PF2-NHC6-NH2
917699 BocA1V2PF2-NHC10-NH2
917958 BocA1V2PF2-NHPEG1-NH2
916692 BocA1V2PF2-NHPEG3-NH2
Other Notes
Legal Information
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service