All Photos(1)
About This Item
Linear Formula:
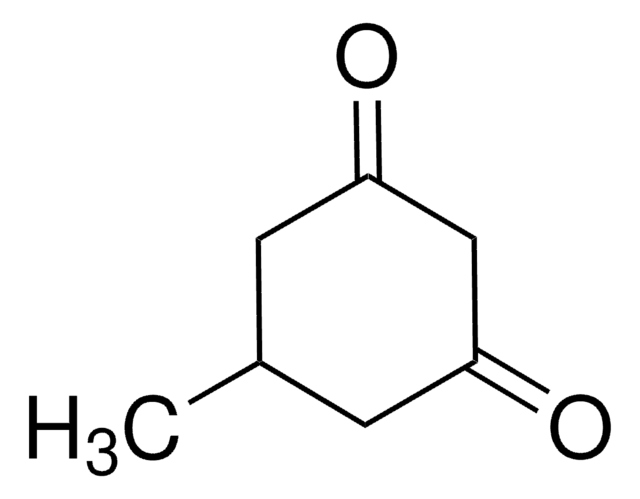
(CH3)2CHC6H7(=O)2
CAS Number:
Molecular Weight:
154.21
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99% (GC)
form
solid
mp
63-65 °C (lit.)
functional group
ketone
shipped in
dry ice
SMILES string
[H]O[H].CC(C)C1CC(=O)CC(=O)C1
InChI
1S/C9H14O2.H2O/c1-6(2)7-3-8(10)5-9(11)4-7;/h6-7H,3-5H2,1-2H3;1H2
InChI key
JYJVWRORYGGPHA-UHFFFAOYSA-N
General description
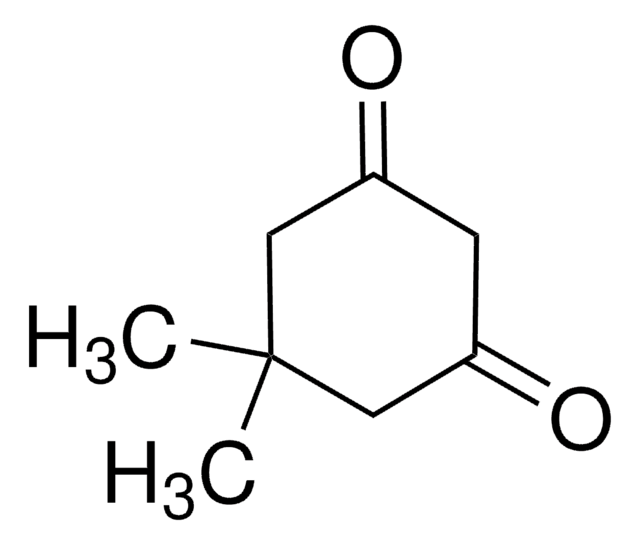
5-Isopropyl-1,3-cyclohexanedione is a cyclic 1,3-diketone. It can be prepared by condensation of isobutylideneacetone with ethyl malonate. 5-Isopropyl-1,3-cyclohexanedione on methylation with methyl iodide yields 5-isopropyl-2-methyl-1,3-cyclohexanedione. A multistep-synthesis of 5-isopropyl-1,3-cyclohexanedione involving aldol condensation, Dieckmann-type annulation, ester hydrolysis and decarboxylation is reported.
Application
5-Isopropyl-1,3-cyclohexanedione may be used as starting reagent for the synthesis of carvotanacetone analogs. It may be used in the preparation of monocyclic terepenes. It may be used in the synthesis of 2H-pyrans, via reaction with α,β-unsaturated aldehydes.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
A Multistep Synthesis Featuring Classic Carbonyl Chemistry for the Advanced Organic Chemistry Laboratory.
Duff DB, et al.
Journal of Chemical Education, 89(3), 406-408 (2011)
Microwave-Assisted Solvent and Catalyst Free Synthesis of 2H-Pyrans.
Edayadulla N and Lee YR.
Bull. Korean Chem. Soc., 34(10), 2963-2963 (2013)
Monocyclic Terpenes from Cyclic 1, 3-Diketones1.
Frank RL and Hall Jr HK.
Journal of the American Chemical Society, 72(4), 1645-1648 (1950)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service