33149
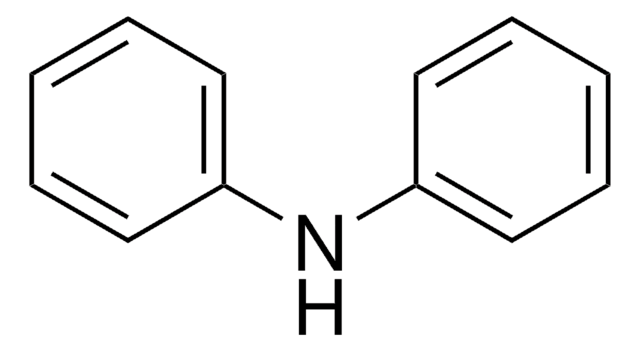
Diphenylamine
puriss. p.a., redox indicator, ACS reagent, reag. Ph. Eur., ≥98% (GC)
About This Item
Recommended Products
grade
ACS reagent
redox indicator
puriss. p.a.
Quality Level
Agency
reag. Ph. Eur.
vapor density
5.82 (vs air)
vapor pressure
1 mmHg ( 108 °C)
Assay
≥98% (GC)
autoignition temp.
1175 °F
technique(s)
titration: suitable
impurities
≤0.01% insoluble in ethanol
ign. residue
≤0.03% (as SO4)
bp
302 °C (lit.)
mp
50-53 °C (lit.)
52.5-54.0 °C
anion traces
nitrate (NO3-): in accordance
cation traces
Fe: ≤10 mg/kg
suitability
suitable for redox indicator
SMILES string
N(c1ccccc1)c2ccccc2
InChI
1S/C12H11N/c1-3-7-11(8-4-1)13-12-9-5-2-6-10-12/h1-10,13H
InChI key
DMBHHRLKUKUOEG-UHFFFAOYSA-N
Gene Information
human ... UGT1A4(54657)
Looking for similar products? Visit Product Comparison Guide
General description
Application
Packaging
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Target Organs
Kidney,Liver,spleen
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Chlorobenzilate; 4-Aminobiphenyl; 2-Fluorobiphenyl; N-Nitrosopyrrolidine; 1,2,4,5-Tetrachlorobenzene; 3-Methylcholanthrene; Phenacetin
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service