543551
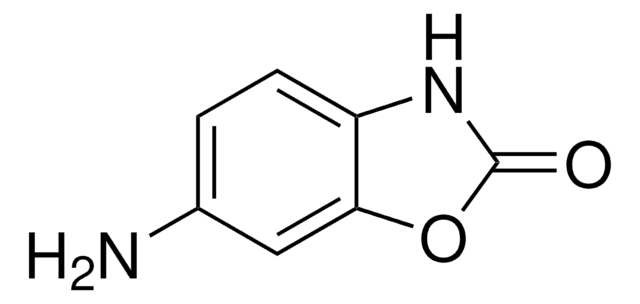
6-Methoxy-2-benzoxazolinone
97%
Synonym(s):
6-MBOA, 6-Methoxy-2(3H)-benzoxazolone, 6-Methoxy-3H-benzoxazol-2-one, 6-Methoxybenzo[d]oxazol-2(3H)-one, 6-Methoxybenzoxazolin-2(3H)-one, 6-Methoxybenzoxazolinone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C8H7NO3
CAS Number:
Molecular Weight:
165.15
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
151-156 °C (lit.)
SMILES string
COc1ccc2NC(=O)Oc2c1
InChI
1S/C8H7NO3/c1-11-5-2-3-6-7(4-5)12-8(10)9-6/h2-4H,1H3,(H,9,10)
InChI key
MKMCJLMBVKHUMS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
6-Methoxy-2-benzoxazolinone (MBOA) is obtained by reacting 2-amino-5-methoxyphenol hydrochloride with urea. MOBA is a naturally occurring auxin-inhibiting substance present in maize shoots.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Structure-activity relationships of benzoxazolinones with respect to auxin-induced growth and auxin-binding protein.
Hoshi-Sakoda, Masako, et al.
Phytochemistry, 37(2), 297-300 (1994)
Hwa Yeon Kim et al.
BMC complementary and alternative medicine, 18(1), 100-100 (2018-03-21)
It is well known that endoplasmic reticulum (ER) stress plays a huge role in development of metabolic diseases. Specially, ER stress-induced cellular dysfunction has a significant involvement in the pathogenesis of human chronic disorders. This study was designed to study
J Acharya et al.
Plant disease, 105(4), 752-757 (2020-10-14)
Corn yield reduction following a cereal rye cover crop has been attributed to, among other factors, allelochemicals released from decomposing cereal rye residue. The allelopathic effect of 6-methoxy-2-benzoxazolinone (MBOA) was evaluated on corn seedling growth, mycelial growth of seven pathogenic
On the Synthesis of 6-Methoxy-2-benzoxazolinone.
John DR, et al.
Agricultural and Biological Chemistry, 39(3), 683-685 (1975)
Thomas Etzerodt et al.
Journal of environmental science and health. Part. B, Pesticides, food contaminants, and agricultural wastes, 43(1), 1-7 (2007-12-29)
Wheat (Triticum aestivum L.) and other cereals produce allelochemicals as natural defense compounds against weeds, fungi, insects and soil-borne diseases. The main benzoxazinoid allelochemical of wheat is 2,4-dihydroxy-7-methoxy-1,4-benzoxazin-3-one (DIMBOA), bound as beta-glucoside and released upon plant injury. When leached from
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service