268402
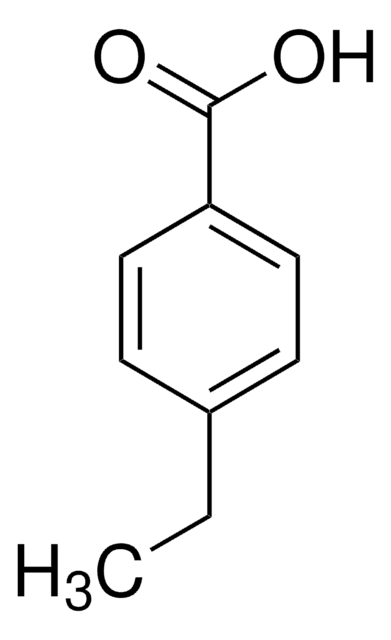
4-Isopropylbenzoic acid
≥96%
Synonym(s):
Cumic acid, Cuminic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
(CH3)2CHC6H4CO2H
CAS Number:
Molecular Weight:
164.20
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥96%
mp
117-120 °C (lit.)
solubility
alcohol: soluble(lit.)
diethyl ether: soluble(lit.)
sulfuric acid: soluble(lit.)
water: slightly soluble(lit.)
SMILES string
CC(C)c1ccc(cc1)C(O)=O
InChI
1S/C10H12O2/c1-7(2)8-3-5-9(6-4-8)10(11)12/h3-7H,1-2H3,(H,11,12)
InChI key
CKMXAIVXVKGGFM-UHFFFAOYSA-N
Related Categories
Application
4-Isopropylbenzoic acid (cuminic acid) was used in the synthesis of three new triorganotin carboxylates bearing methyl, butyl and phenyl substituents at tin, respectively. The products were fully characterized by spectroscopic and thermal techniques, with regard to the coordination number of tin atom, in solution and in the solid state.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Bruno Gaillet et al.
Biotechnology and bioengineering, 106(2), 203-215 (2010-02-24)
Fast and efficient production of recombinant proteins for structural and functional studies is a crucial issue for research and for industry. To this end, we have developed an efficient system to generate in less than 2 months, starting from the
Angiolini L, et al.
Journal of Organometallic Chemistry, 691(9), 1965-1972 (2006)
R W Eaton
Journal of bacteriology, 178(5), 1351-1362 (1996-03-01)
Pseudomonas putida F1 utilizes p-cumate (p-isopropylbenzoate) as a growth substrate by means of an eight-step catabolic pathway. A 35.75-kb DNA segment, within which the cmt operon encoding the catabolism of p-cumate is located, was cloned as four separate overlapping restriction
G J Pass et al.
Xenobiotica; the fate of foreign compounds in biological systems, 32(5), 383-397 (2002-06-18)
1. p-Cymene is an aromatic monoterpene found in the leaves of Eucalyptus spp. and is ingested in the diet of two marsupial folivores, the brushtail possum (Trichosurus vulpecula) and koala (Phascolarctos cinereus). The metabolism of p-cymene by liver microsomes from
L G Puskás et al.
DNA sequence : the journal of DNA sequencing and mapping, 11(1-2), 9-20 (2000-07-21)
Rhodopseudomonas palustris utilizes p-cumate as a carbon source both under anaerobic light and aerobic dark conditions. A gene cluster was isolated whose sequence showed high homology to genes which have been implicated the degradation of p-cumate in Pseudomonas pitida. Seven
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service