144908
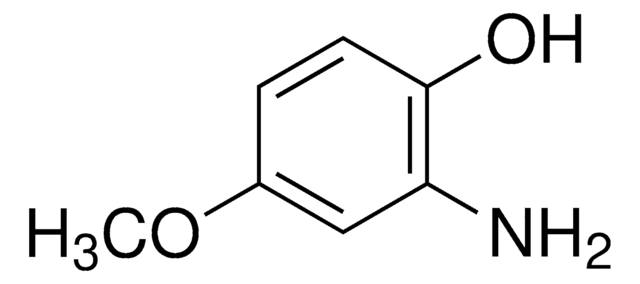
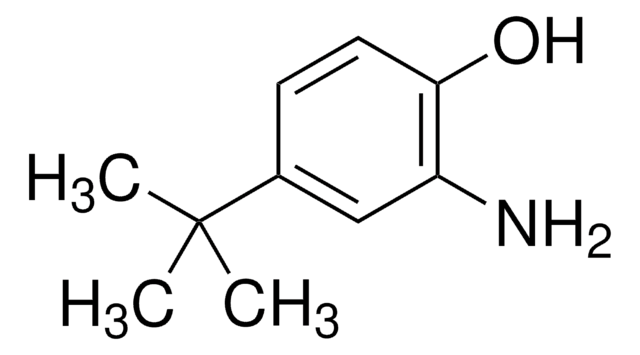
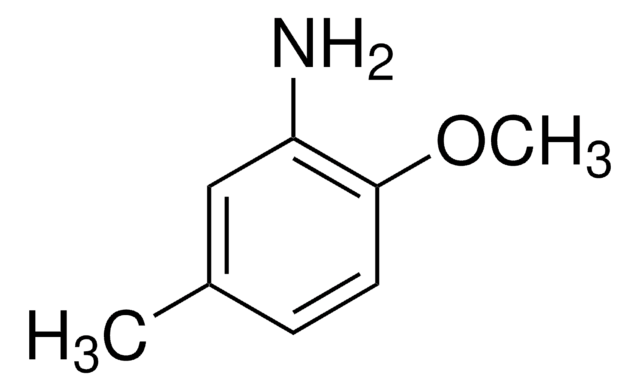
2-Amino-4-methylphenol
97%
Synonym(s):
2-Amino-p-cresol, 2-Hydroxy-5-methylaniline, 3-Amino-4-hydroxytoluene
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
H2NC6H3(CH3)OH
CAS Number:
Molecular Weight:
123.15
Beilstein:
606494
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
mp
133-136 °C (lit.)
SMILES string
Cc1ccc(O)c(N)c1
InChI
1S/C7H9NO/c1-5-2-3-7(9)6(8)4-5/h2-4,9H,8H2,1H3
InChI key
ZMXYNJXDULEQCK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Amino-4-methylphenol is the major sensitizer in contact allergy to Disperse Yellow 3. It reacts with acetylacetone in absolute ethanol to yield 4-(2-hydroxy-5-methylphenyl)imino-2-pentanone. It was converted to dihydrophenoxazinone by purified human hemoglobin.
Application
2-Amino-4-methylphenol was used in the synthesis of novel functionalized spiropyran derivatives of 2H-1,3-benzoxazinone series.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Hongmei Peng et al.
Inorganic chemistry, 47(21), 9828-9835 (2008-10-03)
The synthesis and reactivity of a series of sodium and rare-earth metal complexes stabilized by a dianionic N-aryloxo-functionalized beta-ketoiminate ligand were presented. The reaction of acetylacetone with 1 equiv of 2-amino-4-methylphenol in absolute ethanol gave the compound 4-(2-hydroxy-5-methylphenyl)imino-2-pentanone (LH2, 1)
Antony O Bulanov et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 71(3), 1146-1152 (2008-06-14)
Six novel functionalized spiropyran's derivatives of 2H-1,3-benzoxazinone series were synthesized by introducing the substituents with chelating ability into 2H-chromene part of the 8'-formyl-7'-hydroxy-3-methyl-4-oxo-3,4-dihydro-2H-1,3-benzoxazine-2-spiro-2'-[2H]-chromene (I) by condensation with 2-aminophenol, 2-amino-4-methylphenol, 2-amino-4-nitrophenol, 2-amino-1-methylbenzimidazole, 4-amino-4H-1,2,4-triazole, N-(4-aminophenyl)acetamide. (1)H NMR, UV/vis, IR spectroscopy combined with
Z He et al.
Applied and environmental microbiology, 66(7), 3010-3015 (2000-07-06)
In spite of the variety of initial reactions, the aerobic biodegradation of aromatic compounds generally yields dihydroxy intermediates for ring cleavage. Recent investigation of the degradation of nitroaromatic compounds revealed that some nitroaromatic compounds are initially converted to 2-aminophenol rather
A Tomoda et al.
Journal of biochemistry, 110(6), 1004-1007 (1991-12-01)
2-Amino-4-methylphenol was converted to a brownish yellow material by the lysates of human erythrocytes or purified human hemoglobin. The reaction proceeded oxidatively, coupled with the oxidation of hemoglobin. The major component of the brownish yellow material produced by oxidative condensation
M Akazawa et al.
The Tohoku journal of experimental medicine, 192(4), 301-312 (2001-04-05)
When human erythrocytes were incubated with o-aminophenol at pH 7.0 at 37 degrees C for 46 hours, intracellular oxyhemoglobin was completely oxidized to methemoglobin during the initial 6 hours, and methemoglobin formed was then reduced to oxyhemoglobin during the following
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service