1A02510
USP
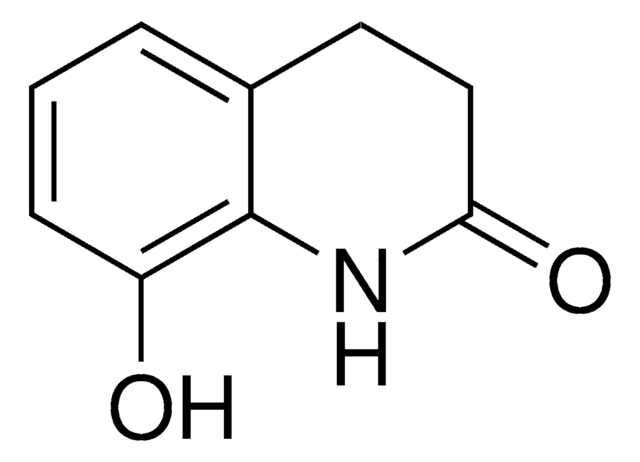
8-Hydroxy-3,4-Dihydroquinolin-2(1H)-One
Pharmaceutical Analytical Impurity (PAI)
Sinónimos:
8-Hydroxy-3,4-dihydroquinolin-2(1H)-one
About This Item
Productos recomendados
grado
pharmaceutical analytical impurity (PAI)
Agency
USP
fabricante / nombre comercial
USP
aplicaciones
pharmaceutical
Formato
neat
temp. de almacenamiento
2-8°C
cadena SMILES
N1c2c(cccc2O)CCC1=O
InChI
1S/C9H9NO2/c11-7-3-1-2-6-4-5-8(12)10-9(6)7/h1-3,11H,4-5H2,(H,10,12)
Clave InChI
UDKMDIKMJWOSJP-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Aripiprazole.
For more information about this PAI, visit here.
Aplicación
Características y beneficios
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
Nota de análisis
Otras notas
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Si necesita más asistencia, póngase en contacto con Atención al cliente
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico