1477003
USP
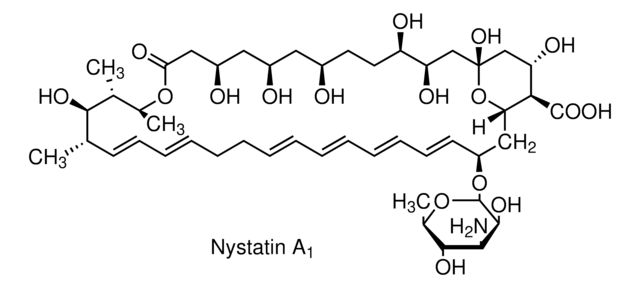
Nystatin
United States Pharmacopeia (USP) Reference Standard
Sinónimos:
Fungicidin, Mycostatin
About This Item
Productos recomendados
grado
pharmaceutical primary standard
familia API
nystatin
fabricante / nombre comercial
USP
aplicaciones
pharmaceutical (small molecule)
formato
neat
cadena SMILES
O[C@@H]([C@H](C)[C@H](C)O1)[C@@H](C)/C=C/C=C/CC/C=C/C=C/C=C/C=C/[C@H](O[C@@H]2O[C@H](C)[C@@H](O)[C@H](N)[C@@H]2O)C[C@@]3([H])[C@H](C(O)=O)[C@@H](O)C[C@](O)(O3)C[C@@H](O)[C@H](O)CC[C@@H](O)C[C@@H](O)C[C@@H](O)CC1=O
InChI
1S/C47H75NO17/c1-27-17-15-13-11-9-7-5-6-8-10-12-14-16-18-34(64-46-44(58)41(48)43(57)30(4)63-46)24-38-40(45(59)60)37(54)26-47(61,65-38)25-36(53)35(52)20-19-31(49)21-32(50)22-33(51)23-39(55)62-29(3)28(2)42(27)56/h5-6,8,10-18,27-38,40-44,46,49-54,56-58,61H,7,9,19-26,48H2,1-4H3,(H,59,60)/b6-5+,10-8+,13-11+,14-12+,17-15+,18-16+/t27-,28+,29-,30+,31+,32+,33+,34-,35+,36+,37-,38-,40+,41-,42+,43+,44-,46-,47+/m0/s1
Clave InChI
VQOXZBDYSJBXMA-QEKUPDCNSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
Also used to prepare standard, and system suitability solution for the assay, composition analysis and content uniformity by using liquid chromatography in conjunction with UV detector according to the given below monographs of United States Pharmacopeia (USP):
- Nystatin, Neomycin Sulfate, Thiostrepton, and Triamcinolone Acetonide Ointment
- Nystatin Oral Suspension
- Nystatin
Acciones bioquímicas o fisiológicas
Antimicrobial spectrum: Nystatin acts against fungi, yeasts and molds.
Precaución
Nota de análisis
Otras notas
Información legal
Producto relacionado
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 2
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico