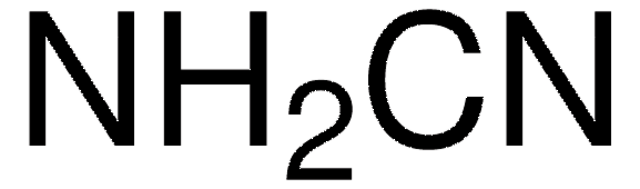H7654
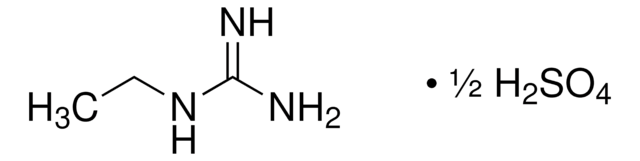
Hydroxyguanidine sulfate salt
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
CH5N3O · 1/2H2SO4
Número de CAS:
Peso molecular:
124.11
Número CE:
Número MDL:
Código UNSPSC:
12352202
ID de la sustancia en PubChem:
NACRES:
NA.77
Productos recomendados
origen biológico
synthetic (organic)
Nivel de calidad
Ensayo
≥98% (TLC)
Formulario
powder
solubilidad
water: 25 mg/mL, clear, colorless
temp. de almacenamiento
2-8°C
cadena SMILES
NC(=N)NO.NC(=N)NO.OS(O)(=O)=O
InChI
1S/2CH5N3O.H2O4S/c2*2-1(3)4-5;1-5(2,3)4/h2*5H,(H4,2,3,4);(H2,1,2,3,4)
Clave InChI
MTGDDPZRXSDPFH-UHFFFAOYSA-N
Acciones bioquímicas o fisiológicas
An early antitumor agent. Oxidation results in release of NO, and formation of other reactive oxygen species, including peroxynitrite and peroxyl radicals. Reacts with NO to form an adduct which is a potent and stable vasodilator.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Rémy Ricoux et al.
European journal of biochemistry, 270(1), 47-55 (2002-12-21)
Nitric oxide (NO) is a potent intra- and intercellular messenger involved in the control of vascular tone, neuronal signalling and host response to infection. In mammals, NO is synthesized by oxidation of l-arginine catalysed by hemeproteins called NO-synthases with intermediate
Ming Xian et al.
Bioorganic & medicinal chemistry, 10(9), 3049-3055 (2002-07-12)
Enzymatic generation of nitric oxide (NO) by nitric oxide synthase (NOS) consists of two oxidation steps. The first step converts L-arginine to N(G)-hydroxy-L-arginine (NOHA), a key intermediate, and the second step converts NOHA to NO and L-citrulline. To fully probe
Tingwei Cai et al.
Bioorganic & medicinal chemistry letters, 12(11), 1507-1510 (2002-05-29)
The electrochemical properties of a series of N-substituted-N'-hydroxyguanidines were studied. Two oxidation potentials of each compound were obtained by cyclic voltammetry. The E(ox1) values were from 0.51 to 0.62V, while the E(ox2) values were from 1.14 to 1.81V in acetonitrile
David Lefèvre-Groboillot et al.
Biochemistry, 42(13), 3858-3867 (2003-04-02)
The interaction of various N-alkyl- and N-aryl-N'-hydroxyguanidines with recombinant NOS containing or not containing tetrahydrobiopterin (BH(4)) was studied by visible, electronic paramagnetic resonance (EPR), and resonance Raman (RR) spectroscopy. N-Hydroxyguanidines interact with the oxygenase domain of BH(4)-free inducible NOS (BH(4)-free
S A Everett et al.
Free radical biology & medicine, 24(1), 1-10 (1998-01-22)
The oxidative denitrification of the antitumour agent hydroxyguanidine (HOG) has been investigated by radiolysis methods and EPR spectroscopy. The azide radical (N3.), a model one-electron oxidant, reacts with HOG with the rate constant 5.1 x 10(9) dm3 mol(-1) s(-1) to
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico