93750
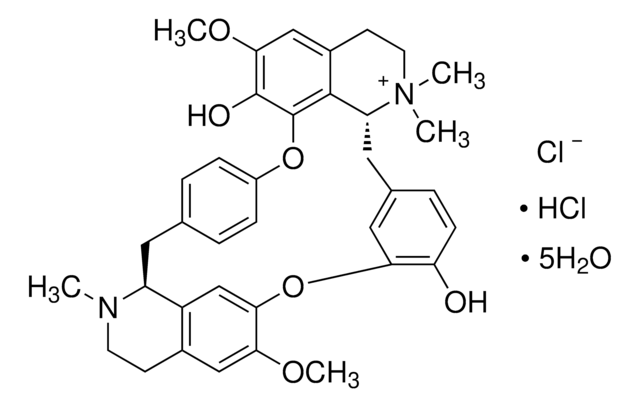
(+)-Tubocurarine chloride pentahydrate
≥97.0% (TLC)
Sinónimos:
(+)-Tubocurarine chloride hydrochloride pentahydrate, D-Tubocurarine dichloride pentahydrate, Tubarine pentahydrate
About This Item
Productos recomendados
origen biológico
plant
Análisis
≥97.0% (TLC)
formulario
powder
actividad óptica
[α]20/D +195±5°, c = 0.5% in H2O
impurezas
~12% water
mp
275-280 °C (dec.) (lit.)
temp. de almacenamiento
2-8°C
cadena SMILES
[Cl-].Cl[H].[H]O[H].[H]O[H].[H]O[H].[H]O[H].[H]O[H].COc1cc2CCN(C)[C@H]3Cc4ccc(Oc5c(O)c(OC)cc6CC[N+](C)(C)[C@H](Cc7ccc(O)c(Oc1cc23)c7)c56)cc4
InChI
1S/C37H40N2O6.2ClH.5H2O/c1-38-14-12-24-19-32(42-4)33-21-27(24)28(38)16-22-6-9-26(10-7-22)44-37-35-25(20-34(43-5)36(37)41)13-15-39(2,3)29(35)17-23-8-11-30(40)31(18-23)45-33;;;;;;;/h6-11,18-21,28-29H,12-17H2,1-5H3,(H-,40,41);2*1H;5*1H2/t28-,29+;;;;;;;/m0......./s1
Clave InChI
WMIZITXEJNQAQK-GGDSLZADSA-N
Información sobre el gen
human ... CHRNA1(1134) , CHRNB1(1140) , CHRND(1144) , CHRNE(1145) , CHRNG(1146)
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
- as an acetylcholine receptor antagonist in synapse blocking experiment to study its effect on neuromuscular junction formation (NMJ) formation in a co-culture system of human skeletal muscles and human stem cell-derived motoneurons
- to induce paralysis in zebrafish larvae to study whole-brain imaging of seizures by two-photon light-sheet microscopy
- to block NMJ to study its role in myotube contraction
Acciones bioquímicas o fisiológicas
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 3 Oral
Código de clase de almacenamiento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico