431311
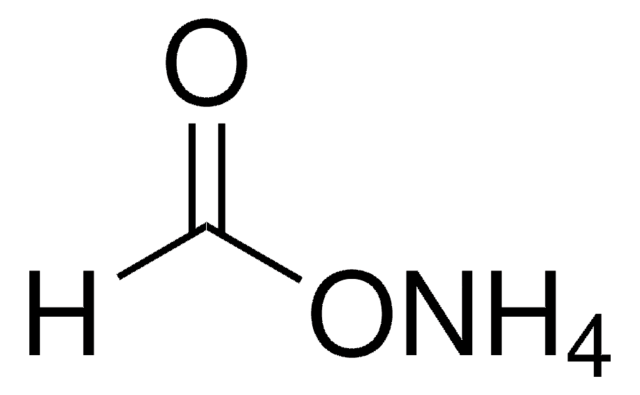
Ammonium acetate
for inorganic trace analysis, ≥99.99% trace metals basis
About This Item
Productos recomendados
grado
for inorganic trace analysis
Agency
suitable for ASTM® 7969
suitable for ASTM® 7979
suitable for DIN 38407-43
suitable for EPA 1634
suitable for EPA 534
suitable for EPA 538.1
suitable for EPA 8328
suitable for EPA ACB B21-03
suitable for EPA ACB B23-05b
suitable for GB 31604.35-2016
suitable for GB 5009.253-2016
suitable for ISO 21675 2019
suitable for ISO 25101
suitable for ISO/CEN 15968-2011
cumplimiento norm.
suitable for FDA C-010.02
presión de vapor
<0.001 hPa
Ensayo
≥99.99% trace metals basis
Formulario
solid
impurezas
≤0.005% insolubles
<100 ppm total metallic impurities
pH
6.7-7.3 (25 °C, 5% in water)
6.7-7.3
mp
110-112 °C (dec.) (lit.)
solubilidad
water: soluble 1,480 g/L at 4 °C
trazas de anión
chloride (Cl-): ≤5 ppm
nitrate (NO3-): ≤0.001%
sulfate (SO42-): ≤0.001%
trazas de catión
Fe: ≤5 ppm
heavy metals: ≤5 ppm
cadena SMILES
N.CC(O)=O
InChI
1S/C2H4O2.H3N/c1-2(3)4;/h1H3,(H,3,4);1H3
Clave InChI
USFZMSVCRYTOJT-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
It may be used in the synthesis of the following:
- Phenanthroline derivatives employed as corrosion inhibitors.
- Highly substituted imidazoles.
- 2,4-Diarylpolyhydroquinoline derivatives.
Información legal
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 1
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico