424633
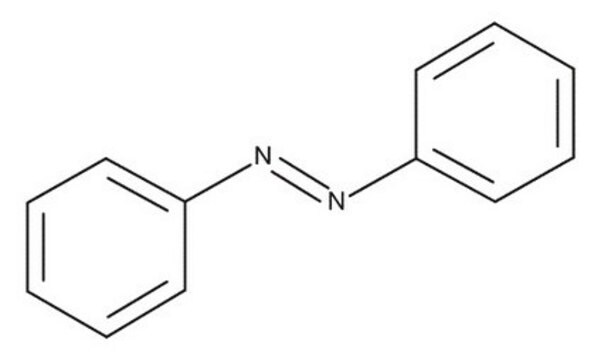
Azobenzene
98%
Sinónimos:
1,2-Diphenyldiazene; Diphenyldiazene
About This Item
Productos recomendados
presión de vapor
1 mmHg ( 104 °C)
Nivel de calidad
Ensayo
98%
Formulario
powder or crystals
temp. de autoignición
890 °F
técnicas
titration: suitable
bp
293 °C (lit.)
mp
65-68 °C (lit.)
densidad
1.09 g/mL at 25 °C (lit.)
aplicaciones
diagnostic assay manufacturing
hematology
histology
temp. de almacenamiento
room temp
cadena SMILES
c1ccc(cc1)\N=N\c2ccccc2
InChI
1S/C12H10N2/c1-3-7-11(8-4-1)13-14-12-9-5-2-6-10-12/h1-10H/b14-13+
Clave InChI
DMLAVOWQYNRWNQ-BUHFOSPRSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
As human tissue is translucent to red and near-infrared light but opaque to blue and UV light, Azobenzene is important in medicine and photopharmacology for applications that involve shifting the absorptions of both trans and cis isomers of azobenzene to longer wavelengths.
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B - Muta. 2 - STOT RE 2
Código de clase de almacenamiento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
212.0 °F - closed cup
Punto de inflamabilidad (°C)
100.0 °C - closed cup
Equipo de protección personal
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Listados normativos
Los listados normativos se proporcionan para los productos químicos principalmente. Para los productos no químicos sólo se puede proporcionar información limitada. Si no hay ninguna entrada, significa que ninguno de los componentes está en la lista. Es obligación del usuario garantizar el uso seguro y legal del producto.
EU REACH Annex XVII (Restriction List)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico