37018
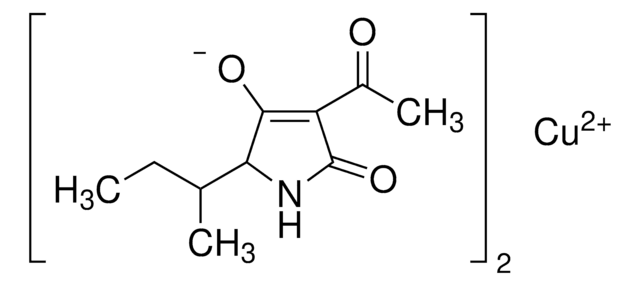
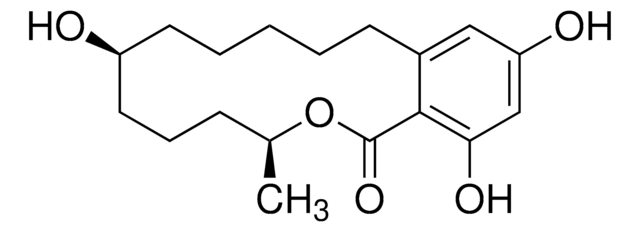
Tenuazonic acid
analytical standard
Sinónimos:
(5S)-3-Acetyl-1,5-dihydro-4-hydroxy-5-[(1S)-1-methylpropyl]-2H-pyrrol-2-one, (S)-3-Acetyl-5-(S)-sec-butyltetramic acid, (S)-3-Acetyl-5-sec-butyl-4-hydroxy-3-pyrrolin-2-one
About This Item
Productos recomendados
grado
analytical standard
Nivel de calidad
caducidad
limited shelf life, expiry date on the label
técnicas
HPLC: suitable
gas chromatography (GC): suitable
aplicaciones
cleaning products
cosmetics
food and beverages
personal care
Formato
neat
temp. de almacenamiento
−20°C
cadena SMILES
CC[C@H](C)[C@@H]1NC(=O)C(C(C)=O)=C1O
InChI
1S/C10H15NO3/c1-4-5(2)8-9(13)7(6(3)12)10(14)11-8/h5,8,13H,4H2,1-3H3,(H,11,14)/t5-,8-/m0/s1
Clave InChI
CEIZFXOZIQNICU-XNCJUZBTSA-N
Descripción general
Aplicación
- Cornflakes using high-performance liquid chromatography (HPLC) technique.
- Human urine samples using isotope dilution assay method and liquid chromatography coupled to a hybrid triple quadrupole/linear ion trap mass spectrometer.
- Cereals using high-performance liquid chromatography–electrospray ionization ion-trap multistage mass spectrometry (HPLC- ESI ion-trap (IT) MS2). ESI Fourier transform-ion cyclotron resonance tandem mass spectrometry (FTICR-MS2) technique is used for product characterization, post derivatization with 2,4-dinitrophenylhydrazine.
Reconstitución
Nota de análisis
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 3 Oral
Código de clase de almacenamiento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico