342001
Procathepsin K, Human, Recombinant, E. coli
Recombinant, human procathepsin K expressed in E. coli with a methionine residue inserted at amino acid 18 to create a new N-terminal initiation site.
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Productos recomendados
recombinante
expressed in E. coli
Nivel de calidad
Ensayo
≥95% (SDS-PAGE)
Formulario
liquid
actividad específica
≥1000 mU/mg protein
fabricante / nombre comercial
Calbiochem®
condiciones de almacenamiento
OK to freeze
avoid repeated freeze/thaw cycles
Condiciones de envío
wet ice
temp. de almacenamiento
−70°C
Descripción general
If the activated enzyme is not used immediately, it is recommended to add methyl methanthiosulfonate (1 mM final concentration MMTS).
Note: 1 mU = 1 milliunit.
Recombinant, human procathepsin K (amino acids 19-329) (GenBank target symbol = S79895, ACC No. P43235) expressed in E. coli with a methionine residue inserted at amino acid 18 to create a new N-terminal initiation site. Cathepsin K, a member of the papain superfamiliy of cysteine proteinases, plays an important role in osteoclast-mediated bone resorption and collagen degradation. Cathepsin K is synthesized as an inactive proenzyme that is converted to its mature, active form by proteolytic cleavage of the 99 amino acid propeptide domain. Inhibitors of cathepsin K include leupeptin (Cat. No. 108975) (IC50 = 70 nM), E-64 (Cat. No. 324890) (IC50 = 5 nM), and cystatin (Cat. No. 324891 or 324896). Requires activation prior to use.
Recombinant, human procathepsin K (amino acids 19-329) (GenBank target symbol = S79895, ACC No. P43235) expressed in E. coli with a methionine residue inserted at amino acid 18 to create a new N-terminal initiation site. Cathepsin K, a member of the papain superfamily of cysteine proteinases, plays an important role in osteoclast-mediated bone resorption and collagen degradation. Cathepsin K is synthesized as an inactive proenzyme that is converted to its mature, active form by proteolytic cleavage of the 99 amino acid propeptide domain. Inhibitors of cathepsin K include leupeptin (Cat. No. 108975) (IC50 = 70 nM), E-64 (Cat. No. 324890) (IC50 = 5 nM), and cystatin (Cat. No. 324891 or 324896). Requires activation prior to use.
Envase
Please refer to vial label for lot-specific concentration.
Advertencia
Toxicity: Standard Handling (A)
Definición de unidad
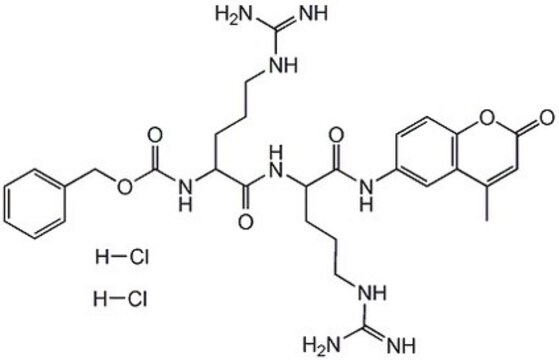
One unit is defined as the amount of enzyme that will hydrolyze 1 µmole benzyloxycarbonyl-phenylalanine-arginine-7-amido-4-methylcoumarin per min at 37°C, pH 5.5.
Forma física
In 500 mM NaCl, 25 mM Tris, pH 8.0.
Reconstitución
Following initial thaw, aliquot and freeze (-70°C). Following activation the enzyme is unstable and should include MMTS for storage (see recommended reaction conditions for activation).
Otras notas
McQueney, M., et al. 1997. J. Bio. Chem.272, 13955.
Bossard, M., et al. 1996. J. Biol. Chem.271, 12517.
Drake, F., et al. 1996. J. Biol. Chem.271, 12511.
Bromme, D. and Okamoto, K. 1995. Biol. Chem. Hoppe-Seyler376, 379.
Baron, R. 1989 Anat. Rec.224, 317.
Littlewood-Evans, A.J., et al. 1975. Cancer Res.57, 5386.
Nishimura, J.S. et al. 1975. Arch. Biochem. Biophys.170, 461.
Bossard, M., et al. 1996. J. Biol. Chem.271, 12517.
Drake, F., et al. 1996. J. Biol. Chem.271, 12511.
Bromme, D. and Okamoto, K. 1995. Biol. Chem. Hoppe-Seyler376, 379.
Baron, R. 1989 Anat. Rec.224, 317.
Littlewood-Evans, A.J., et al. 1975. Cancer Res.57, 5386.
Nishimura, J.S. et al. 1975. Arch. Biochem. Biophys.170, 461.
Información legal
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
Código de clase de almacenamiento
12 - Non Combustible Liquids
Clase de riesgo para el agua (WGK)
WGK 2
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico