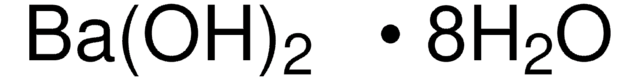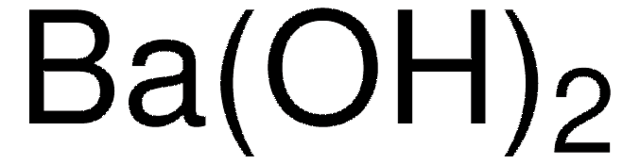1.01735
Barium hydroxide octahydrate
EMPLURA®
Sinónimos:
Barium hydroxide octahydrate, Caustic baryta, Barium oxide hydrate octahydrate
About This Item
Productos recomendados
Nivel de calidad
Línea del producto
EMPLURA®
Ensayo
≥98.0% (acidimetric)
Formulario
solid
potencia
550 mg/kg LD50, oral (Rat)
impurezas
≤0.01% Substances insoluble in dilute hydrochloric acid
≤0.2% Substances not precipitated by dilute sulfuric acid (as sulfate)
pH
14 (20 °C in H2O, saturated aqueous solution)
mp
78 °C
solubilidad
72 g/L
densidad
2.18 g/cm3 at 20 °C
densidad aparente
900‑1100 kg/m3
trazas de anión
carbonate (as BaCO3): ≤2.0%
chloride (Cl-): ≤0.002%
sulfide (S2-): ≤0.001%
trazas de catión
Ca: ≤0.005%
Fe: ≤0.001%
Sr: ≤1.0%
heavy metals (as Pb): ≤0.002%
temp. de almacenamiento
2-30°C
InChI
1S/Ba.10H2O/h;10*1H2/q+2;;;;;;;;;;/p-2
Clave InChI
ZUDYPQRUOYEARG-UHFFFAOYSA-L
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
- Chitosan and carbon nitride doped barium hydroxide nanoparticles served as dye degrader and bactericidal potential: A molecular docking study.: This study explores the use of barium hydroxide octahydrate nanoparticles doped with chitosan and carbon nitride for applications in dye degradation and as bactericidal agents. The research highlights the potential of these nanoparticles in environmental remediation and antimicrobial treatments (Ikram et al., 2023).
- Ba(OH)2 Equilibria in the System Ba-O-H-F, With Application to the Formation of Ba2YCu3O6.5 + x From BaF2-Precursors.: This paper discusses the equilibrium of barium hydroxide octahydrate in the Ba-O-H-F system and its application in forming Ba2YCu3O6.5 + x from BaF2 precursors. This research is relevant to material synthesis and applications in superconductors (Cook et al., 2005).
- The Effect of Nanometer Oxide Additive on the Heat-Conducting Property of Barium Hydroxide Octahydrate.: This article investigates how nanometer-scale oxide additives affect the heat-conducting properties of barium hydroxide octahydrate, highlighting potential applications in thermal management systems and heat storage (Xu et al., 2015).
- Application of capillary isotachophoresis to analysis of serum protein.: This study utilizes barium hydroxide octahydrate in the capillary isotachophoresis analysis of serum proteins, demonstrating its application in biochemical analysis and clinical diagnostics (Ono et al., 1990).
- Staining of tissue sections for electron microscopy with heavy metals. II. Application of solutions containing lead and barium.: This research applies barium hydroxide octahydrate solutions in staining tissue sections for electron microscopy, providing insights into its use in histological and cytological studies (WATSON, 1958).
Nota de análisis
Substances insoluble in dilute hydrochloric acid: ≤ 0.01 %
Carbonate (as BaCO₃): ≤ 2.0 %
Chloride (Cl): ≤ 0.002 %
Sulphide (S): ≤ 0.001 %
Heavy metals (as Pb): ≤ 0.002 %
Ca (Calcium): ≤ 0.005 %
Fe (Iron): ≤ 0.001 %
Sr (Strontium): ≤ 1.0 %
Substances not precipitated by dilute sulfuric acid (as sulfate): ≤ 0.2 %
Información legal
Palabra de señalización
Danger
Frases de peligro
Clasificaciones de peligro
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Skin Corr. 1B
Código de clase de almacenamiento
6.1A - Combustible, acute toxic Cat. 1 and 2 / very toxic hazardous materials
Clase de riesgo para el agua (WGK)
WGK 1
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico