W237809
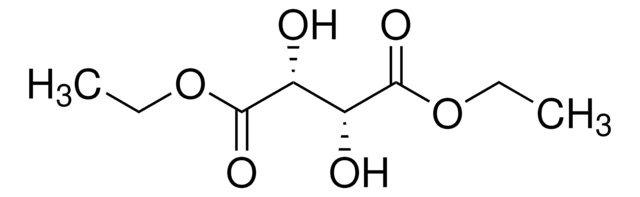
Diethyl L-tartrate
≥99%, FG
Sinónimos:
(+)-Diethyl L-tartrate, L-(+)-Tartaric acid diethyl ester
About This Item
Productos recomendados
origen biológico
synthetic
Nivel de calidad
grado
FG
Fragrance grade
Kosher
Agency
follows IFRA guidelines
meets purity specifications of JECFA
cumplimiento norm.
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
FDA 21 CFR 117
FDA 21 CFR 172.515
Ensayo
≥99%
actividad óptica
[α]20/D +8.5°, neat
índice de refracción
n20/D 1.446 (lit.)
bp
280 °C (lit.)
densidad
1.204 g/mL at 25 °C (lit.)
aplicaciones
flavors and fragrances
Documentación
see Safety & Documentation for available documents
alérgeno alimentario
no known allergens
alérgeno de la fragancia
no known allergens
Organoléptico
fruity; wine-like
cadena SMILES
CCOC(=O)[C@H](O)[C@@H](O)C(=O)OCC
InChI
1S/C8H14O6/c1-3-13-7(11)5(9)6(10)8(12)14-4-2/h5-6,9-10H,3-4H2,1-2H3/t5-,6-/m1/s1
Clave InChI
YSAVZVORKRDODB-PHDIDXHHSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
- Synthesis of l-threitol-based crown ethers and their application as enantioselective phase transfer catalyst in Michael additions.: This study synthesizes l-threitol-based crown ethers using diethyl ʟ-tartrate and explores their efficacy as enantioselective phase transfer catalysts in Michael additions, highlighting their potential in asymmetric synthesis (Rapi et al., 2017).
- A facile approach for the synthesis of C13-C24 fragments of maltepolides A, C and D.: This research demonstrates a novel synthesis method for C13-C24 fragments of maltepolides A, C, and D using diethyl ʟ-tartrate, facilitating the study and development of these bioactive compounds (Rao & Srihari, 2016).
- Development of diacyltetrol lipids as activators for the C1 domain of protein kinase C.: This research introduces diacyltetrol lipids synthesized from diethyl ʟ-tartrate, which act as activators for the C1 domain of protein kinase C, offering insights into signal transduction and therapeutic applications (Mamidi et al., 2012).
- Total synthesis of broussonetine F: the orthoamide Overman rearrangement of an allylic diol.: The paper presents the total synthesis of broussonetine F, utilizing diethyl ʟ-tartrate in an orthoamide Overman rearrangement, showcasing a novel synthetic route for complex natural products (Hama et al., 2011).
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2
Código de clase de almacenamiento
10 - Combustible liquids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
199.4 °F - closed cup
Punto de inflamabilidad (°C)
93 °C - closed cup
Equipo de protección personal
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico