803367
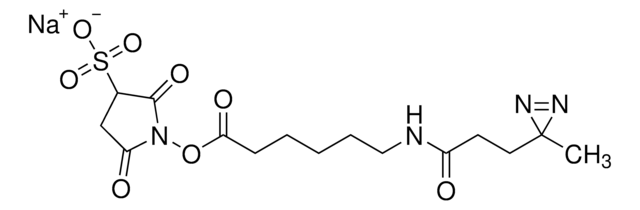
Sulfo-SDAD (Sulfo-NHS-SS-Diazirine) (sulfosuccinimidyl 2-[(4,4′-azipentanamido)ethyl]-1,3′-dithiopropionate]
About This Item
Productos recomendados
Análisis
≥80%
formulario
powder
mol peso
490.51
idoneidad de la reacción
reagent type: cross-linking reagent
condiciones de almacenamiento
desiccated
solubilidad
water: soluble
Condiciones de envío
ambient
temp. de almacenamiento
2-8°C
cadena SMILES
CC1(N=N1)CCC(NCCSSCCC(ON2C(C(S([O-])(=O)=O)CC2=O)=O)=O)=O.[Na+]
InChI
1S/C14H20N4O8S3.Na/c1-14(16-17-14)4-2-10(19)15-5-7-28-27-6-3-12(21)26-18-11(20)8-9(13(18)22)29(23,24)25;/h9H,2-8H2,1H3,(H,15,19)(H,23,24,25);/q;+1/p-1
Clave InChI
BTBHSSBEJHAIAF-UHFFFAOYSA-M
Descripción general
Características y beneficios
- Water soluble—solubility in aqueous solutions improved by a sulfonate group
- Heterobifunctional—NHS ester group reacts with primary amines at pH 7 to 9 to form covalent amide bonds; diazirine (azipentanoate) group reacts efficiently with any amino acid side chain or peptide backbone upon activation with long-wave UV light (330-370 nm)
- Controllable—two-step chemical crosslinking is activated using common laboratory UV lamps
- Easy to use—these crosslinkers are photo-stable under typical laboratory lighting conditions so there is no need to perform experiments in the dark
- Better than aryl azides—the diazirine photoreactive group has better photostability in normal light than phenyl azide groups of traditional photoreactive crosslinkers, yet the diazirine group is more efficiently activated by long-wave UV light
Precaución
Producto relacionado
Código de clase de almacenamiento
13 - Non Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico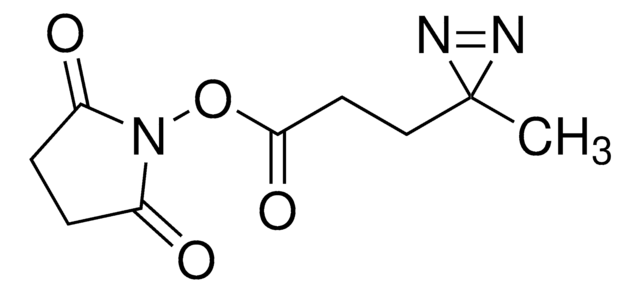
![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)