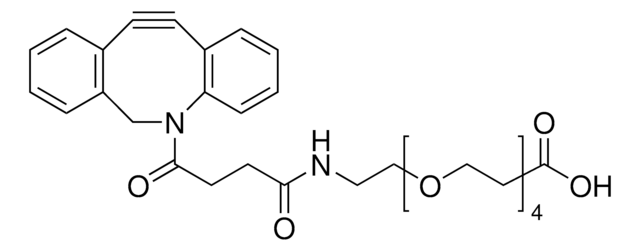
761540
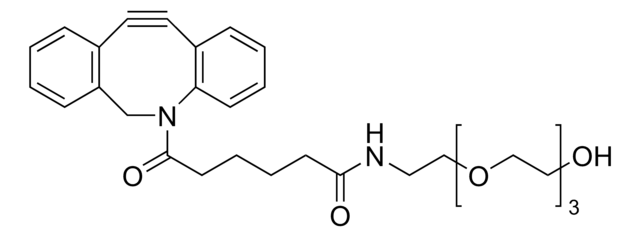
Dibenzocyclooctyne-amine
for Copper-free Click Chemistry
Sinónimos:
DBCO-NH2, DBCO-amine
About This Item
Productos recomendados
formulario
solid
idoneidad de la reacción
reaction type: click chemistry
reagent type: linker
mp
86-96 °C
grupo funcional
amine
temp. de almacenamiento
−20°C
cadena SMILES
NCCC(N1CC2=C(C=CC=C2)C#CC3=C1C=CC=C3)=O
InChI
1S/C18H16N2O/c19-12-11-18(21)20-13-16-7-2-1-5-14(16)9-10-15-6-3-4-8-17(15)20/h1-8H,11-13,19H2
Clave InChI
OCCYFTDHSHTFER-UHFFFAOYSA-N
Descripción general
Aplicación
- Linker to link azide-containing functional groups. It facilitates the formation of linkages through Strain-promoted azide-alkyne cycloaddition reactions (SPAAC)
- Reagent in the synthesis of N-heterocyclic carbene metal thiolates. It functionalizes the metal complexes and increases their reactivity in the strain-promoted alkyne–azide cycloaddition (SPAAC) reactions
- Amine functionalized cyclooctyne derivative. Cyclooctynes are useful in strain-promoted copper-free azide-alkyne cycloaddition reactions. This dibenzocyclooctyne will react with azide functionalized compounds or biomolecules without the need for a Cu(I) catalyst to result in a stable triazole linkage.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico
![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyl N-succinimidyl carbonate for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/969/022/d6776082-2f7a-47c7-bcd4-3830dac0fb7d/640/d6776082-2f7a-47c7-bcd4-3830dac0fb7d.png)
![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethanol for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/171/632/0556139a-2db5-4678-a6ec-a26a693fd574/640/0556139a-2db5-4678-a6ec-a26a693fd574.png)
![N-[(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl]-1,8-diamino-3,6-dioxaoctane for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/294/853/c5e47d84-5aee-4797-aa24-604f291171cc/640/c5e47d84-5aee-4797-aa24-604f291171cc.png)