510017
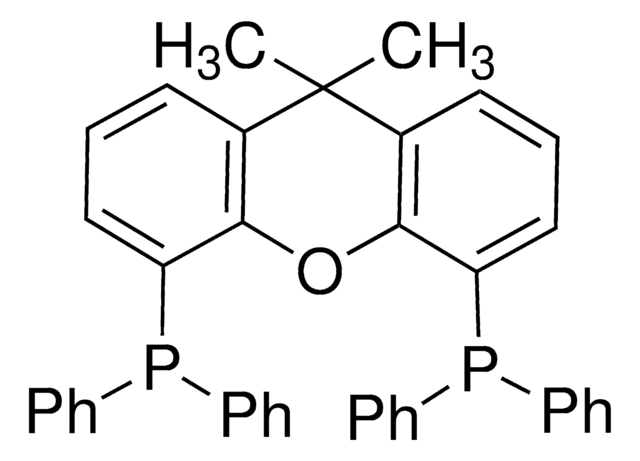
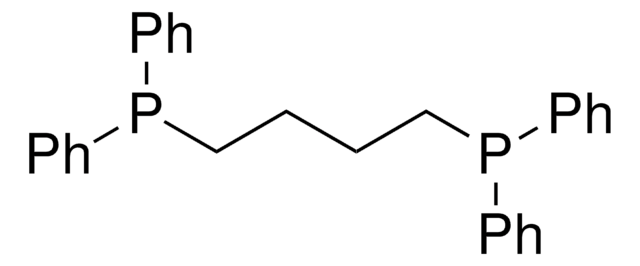
(Oxydi-2,1-phenylene)bis(diphenylphosphine)
98%
Sinónimos:
Bis[(2-diphenylphosphino)phenyl] ether, DPEPhos
About This Item
Productos recomendados
Análisis
98%
idoneidad de la reacción
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: Heck Reaction
reagent type: ligand
reaction type: Hydroaminations
reagent type: ligand
reaction type: Negishi Coupling
reagent type: ligand
reaction type: Suzuki-Miyaura Coupling
características de los productos alternativos más sostenibles
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
184-187 °C (lit.)
grupo funcional
phosphine
categoría alternativa más sostenible
, Aligned
cadena SMILES
O(c1ccccc1P(c2ccccc2)c3ccccc3)c4ccccc4P(c5ccccc5)c6ccccc6
InChI
1S/C36H28OP2/c1-5-17-29(18-6-1)38(30-19-7-2-8-20-30)35-27-15-13-25-33(35)37-34-26-14-16-28-36(34)39(31-21-9-3-10-22-31)32-23-11-4-12-24-32/h1-28H
Clave InChI
RYXZOQOZERSHHQ-UHFFFAOYSA-N
Categorías relacionadas
Descripción general
Aplicación
Ruthenium-Catalyzed N-Alkylation of Amines and Sulfonamides Using Borrowing Hydrogen Methodology
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico