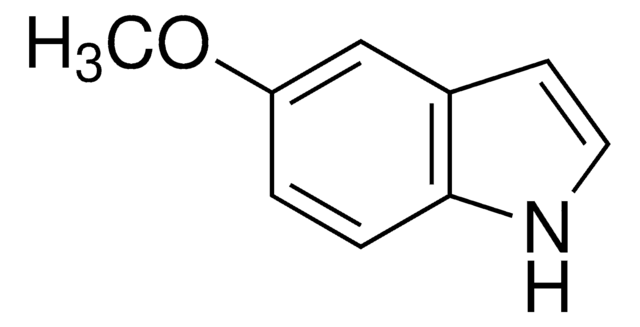
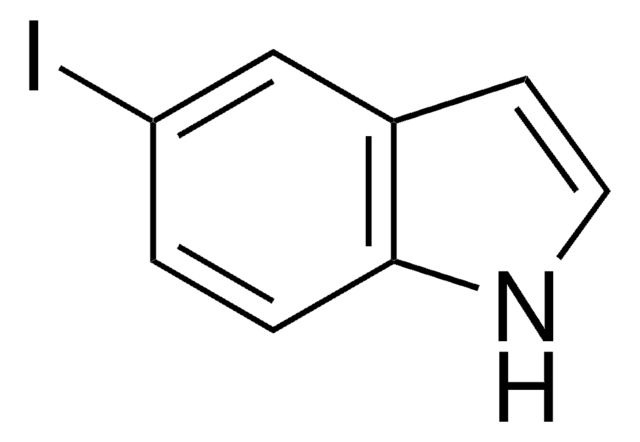
222410
5-Methylindole
99%
Sinónimos:
NSC 522562
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C9H9N
Número de CAS:
Peso molecular:
131.17
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
99%
Formulario
solid
mp
60-62 °C (lit.)
cadena SMILES
Cc1ccc2[nH]ccc2c1
InChI
1S/C9H9N/c1-7-2-3-9-8(6-7)4-5-10-9/h2-6,10H,1H3
Clave InChI
YPKBCLZFIYBSHK-UHFFFAOYSA-N
Descripción general
The binding of 5-methylindole (inducer) to the Escherichia coli trp repressor has been studied. The mass analyzed threshold ionization spectra of jetcooled 5-methylindole (5MI) has also been studied.
Aplicación
Reactant for preparation of:
- Pharmaceutically active 2-oxo-1-pyrrolidine analogues
- Potential anticancer immunomodulators
- Preparation of antifungal agents
- Sodium-dependent glucose co-transporter 2 (SGLT2) inhibitors for the management of hyperglycemia in diabetes
- IL2-inducible T-cell kinase (ITK) inhibitors
- Checkpoint 1 kinase inhibitors
- CRTh2 antagonists
- Inhibitors of human immunodeficiency virus type 1 (HIV-1) attachment
- Agonists of the histamine H4 receptor
- Monoamine reuptake inhibitors
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Changjiang Dong et al.
Science (New York, N.Y.), 309(5744), 2216-2219 (2005-10-01)
Chlorinated natural products include vancomycin and cryptophycin A. Their biosynthesis involves regioselective chlorination by flavin-dependent halogenases. We report the structural characterization of tryptophan 7-halogenase (PrnA), which regioselectively chlorinates tryptophan. Tryptophan and flavin adenine dinucleotide (FAD) are separated by a 10
Jung Lee Lin et al.
The Journal of chemical physics, 120(11), 5057-5063 (2004-07-23)
The vibrationally resolved mass analyzed threshold ionization spectra of jetcooled 5-methylindole (5MI) and 3-methylindole (3MI) have been recorded by ionizing via various vibronic levels of each species. The adiabatic ionization energies (IEs) of 5MI and 3MI are determined to be
P Babitzke et al.
The Journal of biological chemistry, 270(21), 12452-12456 (1995-05-26)
A filter binding assay was used to determine the structural features of L-tryptophan required for activation of TRAP, the trp RNA-binding attenuation protein of Bacillus subtilis. We examined the ability of L-tryptophan and 26 of its analogs to activate TRAP.
F Peter Guengerich et al.
Journal of medicinal chemistry, 47(12), 3236-3241 (2004-05-28)
Indigoids, a class of bis-indoles, represent a promising protein kinase inhibitor scaffold. Oxidation of indole by cytochrome P450 (P450) has been shown to generate species (indoxyl, isatin) that couple to yield indigo and indirubin. Escherichia coli-expressed human P450 2A6 mutants
Dorleta Gonzalez et al.
Bioorganic & medicinal chemistry, 26(9), 2551-2560 (2018-04-17)
Following the premises of the multitarget-directed ligands approach for the drug R&D against neurodegenerative diseases, where Alzheimer's disease (AD) outstands, we have synthesized and evaluated analogues of the gramine derivative ITH12657 (1-benzyl-5-methyl-3-(piperidin-1-ylmethyl-1H-indole, 2), which had shown important neuroprotective properties, such
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico