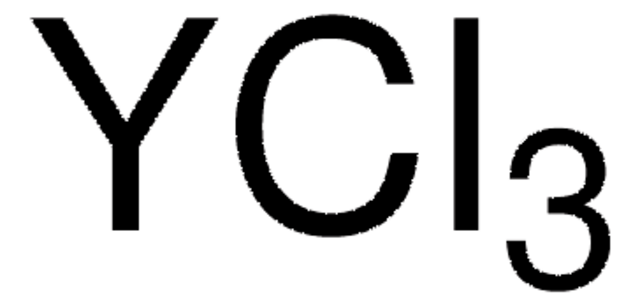211648
Yttrium(III) chloride hexahydrate
99.9% trace metals basis
Sinónimos:
Yttrium trichloride hexahydrate
About This Item
Productos recomendados
Nivel de calidad
Ensayo
99.9% trace metals basis
Formulario
crystals and lumps
impurezas
≤1500.0 ppm Trace Rare Earth Analysis
mp
100 °C (dec.) (lit.)
densidad
2.18 g/mL at 25 °C (lit.)
cadena SMILES
[H]O[H].[H]O[H].[H]O[H].[H]O[H].[H]O[H].[H]O[H].Cl[Y](Cl)Cl
InChI
1S/3ClH.6H2O.Y/h3*1H;6*1H2;/q;;;;;;;;;+3/p-3
Clave InChI
IINACGXCEZNYTF-UHFFFAOYSA-K
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Aplicación
- Yttrium(III) chloride hexahydrate: This compound is extensively used in the synthesis of yttrium-based materials, including phosphors and ceramics. It acts as a precursor for yttrium iron garnets, crucial components in electronic and optical devices due to their magnetic properties. Additionally, its use in catalysis and materials science research is notable, especially where high purity yttrium is required (Sigma-Aldrich, CAS 10025-94-2).
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Sens. 1B
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
The rare earth elements impact nearly everyone in the world. All of the people living in advanced technological countries and almost all those living in third world countries utilize the rare earths in their everyday living—the car that one drives (gasoline is refined from oil using rare earth catalysts and catalytic converters reduce the polluting emissions from the automotive exhaust), watching the news on TV (the red and green colors in TV screens), the telephones and computers we use to communicate (the permanent magnets in speakers and disc drives), just to name a few examples.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico