143685
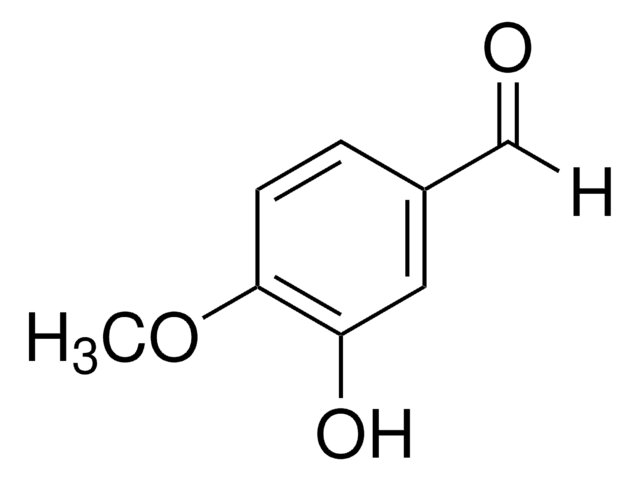
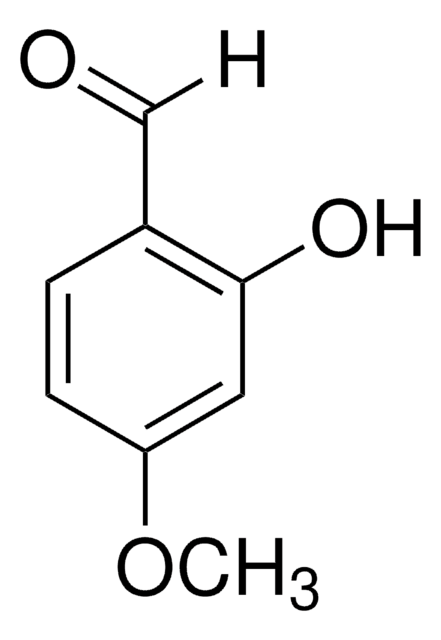
3-Hydroxy-4-methoxybenzaldehyde
99%
Sinónimos:
3-Hydroxyanisaldehyde, Isovanillin
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
HOC6H3(OCH3)CHO
Número de CAS:
Peso molecular:
152.15
Beilstein:
1073021
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Análisis
99%
bp
179 °C/15 mmHg (lit.)
mp
113-115 °C (lit.)
grupo funcional
aldehyde
cadena SMILES
[H]C(=O)c1ccc(OC)c(O)c1
InChI
1S/C8H8O3/c1-11-8-3-2-6(5-9)4-7(8)10/h2-5,10H,1H3
Clave InChI
JVTZFYYHCGSXJV-UHFFFAOYSA-N
Categorías relacionadas
Descripción general
3-Hydroxy-4-methoxybenzaldehyde on condensation with furan-2-carboxylic acid hydrazide and thiophene-2-carboxylic acid hydrazide yields Schiff-bases. It undergoes condensation reaction with1-azabicyclo[2.2.2]octan-3-one to give (Z)-2-(3-hydroxy-4-methoxybenzylidene)-1-azabicyclo[2.2.2]octan-3-one.
Aplicación
3-Hydroxy-4-methoxybenzaldehyde was used as starting reagent during the two-step stereoselective synthesis of the anticancer drug (Z)-combretastatin A-4 and glycitein synthesis.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
>212.0 °F
Punto de inflamabilidad (°C)
> 100 °C
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Caroline Lang'at-Thoruwa et al.
Journal of natural products, 66(1), 149-151 (2003-01-25)
4-Methoxyresorcinol (3) was synthesized as the precursor for glycitein (6) synthesis by the oxidation of 3-hydroxy-4-methoxybenzaldehyde (1) to the aryl formate with H2O2 and a catalytic amount of SeO2. Glycitein (6) was synthesized by cyclization of 2,4,4'-trihydroxy-5-methoxydeoxybenzoin (5) with N,N-dimethylformamide
K Gaukroger et al.
The Journal of organic chemistry, 66(24), 8135-8138 (2001-11-28)
A high-yielding, two-step stereoselective synthesis of the anticancer drug (Z)-combretastatin A-4 (1) has been devised. The method uses the Perkin condensation of 3,4,5-trimethoxyphenylacetic acid and 3-hydroxy-4-methoxybenzaldehyde followed by decarboxylation of the cinnamic acid intermediate using copper and quinoline. The iodine-catalyzed
Vijayakumar N Sonar et al.
Acta crystallographica. Section C, Crystal structure communications, 59(Pt 11), o647-o649 (2003-11-08)
Crystals of the title compound, C(15)H(17)NO(3), were obtained from a condensation reaction of 3-hydroxy-4-methoxybenzaldehyde with 1-azabicyclo[2.2.2]octan-3-one and subsequent crystallization of the product from methanol. The title compound, containing a double bond that connects the azabicyclic ring system to the 3-hydroxy-4-methoxybenzylidene
Riyadh M Ahmed et al.
TheScientificWorldJournal, 2013, 754868-754868 (2013-09-13)
New monomeric cobalt and cadmium complexes with Schiff-bases, namely, N'-[(E)-(3-hydroxy-4-methoxyphenyl)methylidene]furan-2-carbohydrazide (L¹) and N'-[(E)-(3-hydroxy-4-methoxyphenyl)methylidene]thiophene-2-carbohydrazide (L²) are reported. Schiff-base ligands L¹ and L² were derived from condensation of 3-hydroxy-4-methoxybenzaldehyde (iso-vanillin) with furan-2-carboxylic acid hydrazide and thiophene-2-carboxylic acid hydrazide, respectively. Complexes of the
Michael D Markey et al.
Organic letters, 9(17), 3255-3257 (2007-07-31)
The first total synthesis of santiagonamine (1) is achieved in 12 steps from isovanillin. A palladium-catalyzed Ullmann cross-coupling reaction and a photocyclization are the key steps in the synthesis.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico