120227
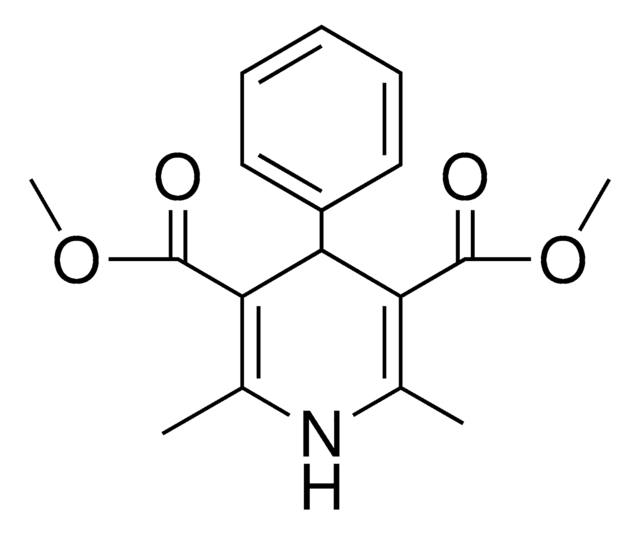
Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate
95%
Sinónimos:
Hantzsch ester
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C13H19NO4
Número de CAS:
Peso molecular:
253.29
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
95%
Formulario
solid
mp
178-183 °C (lit.)
solubilidad
organic solvents: soluble
grupo funcional
ester
cadena SMILES
CCOC(=O)C1=C(C)NC(C)=C(C1)C(=O)OCC
InChI
1S/C13H19NO4/c1-5-17-12(15)10-7-11(13(16)18-6-2)9(4)14-8(10)3/h14H,5-7H2,1-4H3
Clave InChI
LJXTYJXBORAIHX-UHFFFAOYSA-N
Categorías relacionadas
Descripción general
Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate is often used as a building block in organic synthesis for the preparation of various biologically active compounds.
Aplicación
Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate (DTP) was used to study the mechanism of electrochemical oxidation of DTP in ethanol/water solutions on a glassy carbon electrode.
Used as a hydrogen source in organocatalytic reductive amination and conjugate reduction.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 2
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
[Geroprotective activity of 2,6-dimethyl-3,5-diethoxycarbonyl-l,4-dihydropyridine].
N M Emanuél' et al.
Doklady Akademii nauk SSSR, 284(5), 1271-1274 (1985-01-01)
M V Bilenko et al.
Biulleten' eksperimental'noi biologii i meditsiny, 96(9), 8-11 (1983-09-01)
Prophylactic injection of the natural antioxidant alpha-tocopherol and synthetic antioxidants ionol, diludin and 6-mercurascan prevented the development of lesions during acute renal ischemia and subsequent reperfusion. Ionol proved more effective on intraperitoneal injection than on intragastric route of administration. It
M Mamadiev et al.
Voprosy meditsinskoi khimii, 29(2), 83-89 (1983-03-01)
Appearance of cadaverine deaminating activity in mitochondrial fractions of liver and kidney of rabbits with experimental alimentary hypercholesterolaemia was prevented by an antioxant diludin (2,6-dimethyl-3,5-diethoxycarbonyl-1,4-dihydropyridine) which also decreased the abnormally elevated AMP-deaminating activity and elevated the decreased monoamine oxidase activity
Jing Zhang et al.
iScience, 23(1), 100755-100755 (2019-12-31)
The alkoxyl radical is an essential reactive intermediate in mechanistic studies and organic synthesis with hydrogen atom transfer (HAT) reactivity. However, compared with intramolecular 1,5-HAT or intermolecular HAT of alkoxyl radicals, the intramolecular 1,2-HAT reactivity has been limited to theoretical
N A Basova et al.
Rossiiskii fiziologicheskii zhurnal imeni I.M. Sechenova, 88(5), 650-657 (2002-07-26)
The main concern of this work was to examine the relation between altered antioxidant status on the one hand and increase in L-tryptophan absorption in the small intestine in order to bring further information regarding to possible role of vitamin
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico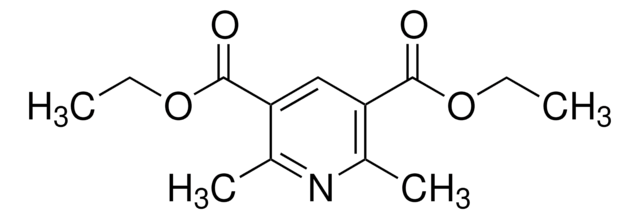


![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)
![Tris[2-phenylpyridinato-C2,N]iridium(III) sublimed grade](/deepweb/assets/sigmaaldrich/product/structures/167/234/658d0b76-d31d-4fd5-8041-e04e207227c9/640/658d0b76-d31d-4fd5-8041-e04e207227c9.png)