558958
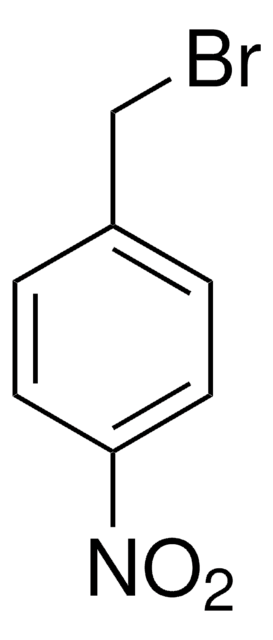
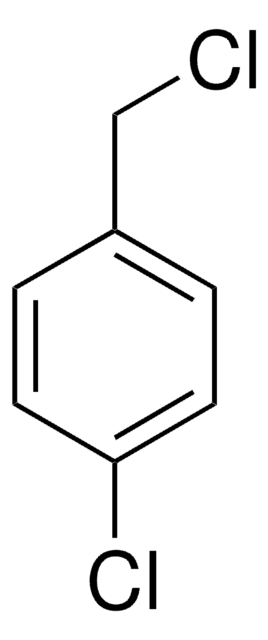
4-Chlorobenzyl bromide
97%
Synonym(s):
α-Bromo-p-chlorotoluene, 1-(Bromomethyl)-4-chlorobenzene, 1-Chloro-4-bromomethylbenzene, p-Chlorobenzyl bromide
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
ClC6H4CH2Br
CAS Number:
Molecular Weight:
205.48
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
mp
48-52 °C (lit.)
SMILES string
Clc1ccc(CBr)cc1
InChI
1S/C7H6BrCl/c8-5-6-1-3-7(9)4-2-6/h1-4H,5H2
InChI key
KQNBRMUBPRGXSL-UHFFFAOYSA-N
General description
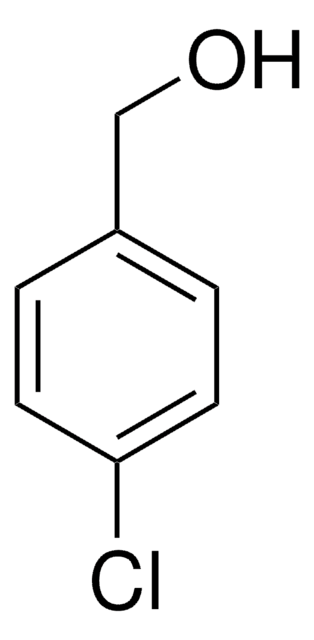
4-Chlorobenzyl bromide undergoes carbonylation in the presence of dimer of chloro(1,5-cyclooctadiene)rhodium(I) to yield the corresponding phenylacetic acid. It can be synthesized by reacting 4-chlorobenzyl alcohol with bromodimethylsulfonium bromide (BDMS) It can also be synthesized by refluxing a mixture of 4-chlorobenzaldehyde, chlorotrimethylsilane, 1,1,3,3-tetramethyldisiloxane and lithium bromide.
Application
4-Chlorobenzyl bromide may be used to synthesize 1-(4-chlorobenzyl)-2-(pyrrolidin-1-yl-methyl)-1H-benzimidazole dihydrochloride
Packaging
4-Chlorobenzyl bromide may be used to synthesize:
- 1-[1-[2-[(4-chlorobenzyl)thio]phenyl]vinyl]-1H-imidazole
- 6-[4-(4-chlorobenzyl)piperazinyl]chromane
- N,N′-bis(4-chlorobenzyl)piperazine
- N-(4-chlorobenzyl)piperazine
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
6-(4-Benzylpiperazin-1-yl) benzodioxanes as selective ligands at cloned primate dopamine D 4 receptors.
Hodgetts KJ, et al.
Bioorganic & Medicinal Chemistry, 9(12), 3207-3213 (2001)
"Reduction of carbonyl compounds promoted by silicon hydrides under the influence of trimethylsilyl-based reagent"
Aizpurua.MJ, et al.
Canadian Journal of Chemistry, 64(12), 2342-2347 (1986)
Gazi S and Ananthakrishnan R
Royal Society of Chemistry Advances, 2(20), 7781- 7787 (2012)
Synthesis and antifungal activity of new 1-vinylimidazoles.
Ogata M, et al.
Journal of Medicinal Chemistry, 30(8), 1348-1354 (1987)
Synthesis of phenylacetic acids under rhodium-catalyzed carbonylation conditions.
Giroux A, et al.
Tetrahedron Letters, 41(40), 7601-7604 (2000)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service