468037
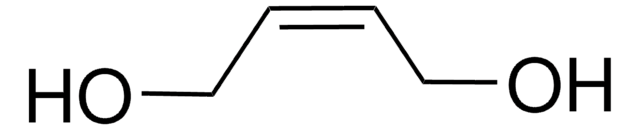
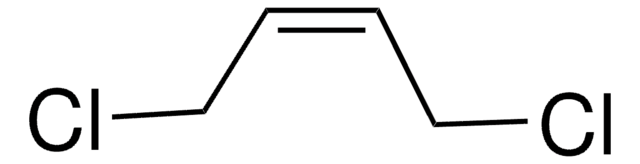
cis-1,4-Diacetoxy-2-butene
95%
Synonym(s):
cis-2-Butene-1,4-diol diacetate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3CO2CH2CH=CHCH2O2CCH3
CAS Number:
Molecular Weight:
172.18
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
refractive index
n20/D 1.443 (lit.)
bp
120-121 °C/18 mmHg (lit.)
density
1.08 g/mL at 25 °C (lit.)
functional group
ester
SMILES string
[H]\C(COC(C)=O)=C(/[H])COC(C)=O
InChI
1S/C8H12O4/c1-7(9)11-5-3-4-6-12-8(2)10/h3-4H,5-6H2,1-2H3/b4-3-
InChI key
VZUAUHWZIKOMFC-ARJAWSKDSA-N
General description
cis-1,4-Diacetoxy-2-butene is an ester. Ruthenium olefin metathesis catalysts having cyclic (alkyl)(amino)carbenes effectively catalyzes the cross-metathesis of cis-1,4-diacetoxy-2-butene with allylbenzene. cis-1,4-Diacetoxy-2-butene on pyrolysis affords 1-acetoxy-1,3-butadiene, a diacetate and an isomeric 1,2-diacetoxy-3-butene. Mechanism of pyrolysis has been investigated.
Application
cis-1,4-Diacetoxy-2-butene may be used in the preparation of hindered Diels-Alder adducts. It may be used for the synthesis of 1,2,3,4-tetrahydro-2-vinylquinoxalines, via Palladium-catalyzed tandem allylation of 1,2-phenylenediamines.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Kerstin Rosmarion et al.
Applied spectroscopy, 59(3), 366-375 (2005-05-20)
Hindered Diels-Alder adducts have been prepared from 3,6-dibromophencyclone, 2, with cis-1,4-diacetoxy-2-butene, 3; cis-2-butene-1,4-diol, 4; and N-benzylmaleimide, 5. The adduct from the parent phencyclone, 1, with N-benzylmaleimide was prepared for comparison. One- and two-dimensional (1D and 2D) proton and carbon-13 NMR
Pyrolysis of Esters. V. Mechanism of 1, 4-Elimination1.
Bailey WJ and BARCLAY Jr R.
The Journal of Organic Chemistry, 21(3), 328-331 (1956)
Donde R Anderson et al.
Organometallics, 27(4), 563-566 (2008-06-28)
The evaluation of ruthenium olefin metathesis catalysts 4-6 bearing cyclic (alkyl)(amino)carbenes (CAACs) in the cross-metathesis of cis-1,4-diacetoxy-2-butene (7) with allylbenzene (8) and the ethenolysis of methyl oleate (11) is reported. Relative to most NHC-substituted complexes, CAAC-substituted catalysts exhibit lower E/Z
Palladium-catalyzed tandem allylation of 1, 2-phenylenediamines with cis-1, 4-diacetoxy-2-butene.
Yang SC, et al.
Tetrahedron Letters, 45(25), 4951-4954 (2004)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service