406147
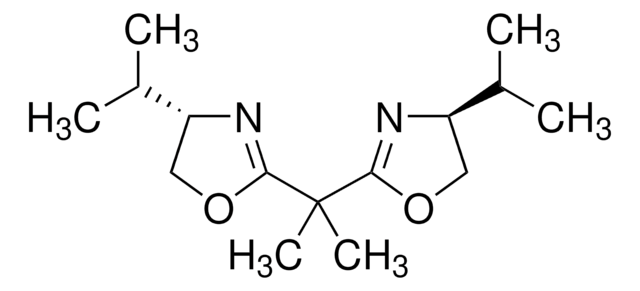
2,2′-Isopropylidenebis[(4S)-4-tert-butyl-2-oxazoline]
99%
Synonym(s):
(S,S)-2,2′-Isopropylidene-bis(4-tert-butyl-2-oxazoline)
About This Item
Recommended Products
Assay
99%
optical activity
[α]20/D −120°, c = 5 in chloroform
mp
89-91 °C (lit.)
functional group
ether
SMILES string
CC(C)(C)[C@H]1COC(=N1)C(C)(C)C2=N[C@H](CO2)C(C)(C)C
InChI
1S/C17H30N2O2/c1-15(2,3)11-9-20-13(18-11)17(7,8)14-19-12(10-21-14)16(4,5)6/h11-12H,9-10H2,1-8H3/t11-,12-/m1/s1
InChI key
DPMGLJUMNRDNMX-VXGBXAGGSA-N
Related Categories
Application
related product
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
C2-symmetric chiral bisoxazolines (BOX) are privileged structures because they promote a great number of transformations with unprecedented selectivity.1
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 406147-50MG | |
| 406147-250MG | 4061833055182 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![(−)-2,2′-Isopropylidenebis[(4S)-4-phenyl-2-oxazoline] 97%](/deepweb/assets/sigmaaldrich/product/structures/297/720/a29f61c3-34e4-410c-acdd-241699b80af3/640/a29f61c3-34e4-410c-acdd-241699b80af3.png)
![2,2′-Bis[(4S)-4-benzyl-2-oxazoline] 98%](/deepweb/assets/sigmaaldrich/product/structures/139/783/42da3c77-52af-401b-8525-35d96415e284/640/42da3c77-52af-401b-8525-35d96415e284.png)
![[3aR-[2(3′aR*,8′aS*),3′aβ,8′aβ]]-(+)-2,2′-Methylenebis[3a,8a-dihydro-8H-indeno[1,2-d]oxazole] 98%](/deepweb/assets/sigmaaldrich/product/structures/134/031/294d2464-1571-4514-8e4c-c0cda1c1df7b/640/294d2464-1571-4514-8e4c-c0cda1c1df7b.png)
![(+)-2,2′-Isopropylidenebis[(4R)-4-phenyl-2-oxazoline] 97%](/deepweb/assets/sigmaaldrich/product/structures/232/241/07f8baaa-0ba2-49e0-8ac2-f6d256fb2c84/640/07f8baaa-0ba2-49e0-8ac2-f6d256fb2c84.png)
![2,2′-Methylenebis[(4S)-4-phenyl-2-oxazoline] 97%](/deepweb/assets/sigmaaldrich/product/structures/255/350/4403d4f8-c973-4da7-a5b6-2e93d1eacb10/640/4403d4f8-c973-4da7-a5b6-2e93d1eacb10.png)
![2,2′-Methylenebis[(4S)-4-tert-butyl-2-oxazoline] 99%](/deepweb/assets/sigmaaldrich/product/structures/316/483/d76fb8bf-5216-440e-a409-771970cad43d/640/d76fb8bf-5216-440e-a409-771970cad43d.png)

![2,6-Bis[(4S)-4-phenyl-2-oxazolinyl]pyridine 98%](/deepweb/assets/sigmaaldrich/product/structures/372/262/fb5c79fe-8277-48b0-a73e-4124c7c2c41c/640/fb5c79fe-8277-48b0-a73e-4124c7c2c41c.png)
![2,6-Bis[(4S)-(−)-isopropyl-2-oxazolin-2-yl]pyridine 99%](/deepweb/assets/sigmaaldrich/product/structures/452/550/7e22a7c6-e84a-4741-af9a-e40f05d8061c/640/7e22a7c6-e84a-4741-af9a-e40f05d8061c.png)
![2,6-Bis[(4R)-4-phenyl-2-oxazolinyl]pyridine 98%](/deepweb/assets/sigmaaldrich/product/structures/888/428/3be8313a-627a-4281-be35-306cc5da562a/640/3be8313a-627a-4281-be35-306cc5da562a.png)
![2,6-Bis[(3aR,8aS)-(+)-8H-indeno[1,2-d]oxazolin-2-yl)pyridine ≥94%](/deepweb/assets/sigmaaldrich/product/structures/123/619/565288e2-e1c9-4825-a440-17e786bc2c27/640/565288e2-e1c9-4825-a440-17e786bc2c27.png)