269107
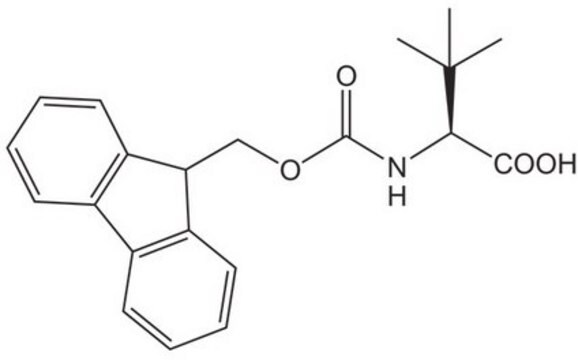
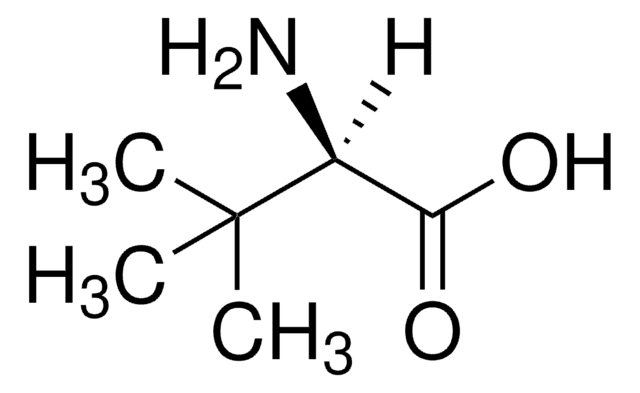
L-tert-Leucine
99%, for peptide synthesis
Synonym(s):
(S)-2-Amino-3,3-dimethylbutyric acid, L-α-tert-Butylglycine
About This Item
Recommended Products
product name
L-tert-Leucine, 99%
Assay
99%
form
powder
optical activity
[α]20/D −9.5°, c = 3 in H2O
optical purity
ee: 99% (GLC)
reaction suitability
reaction type: solution phase peptide synthesis
mp
≥300 °C (lit.)
application(s)
peptide synthesis
SMILES string
CC(C)(C)[C@H](N)C(O)=O
InChI
1S/C6H13NO2/c1-6(2,3)4(7)5(8)9/h4H,7H2,1-3H3,(H,8,9)/t4-/m1/s1
InChI key
NPDBDJFLKKQMCM-SCSAIBSYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- As a key precursor in the synthesis of a chiral phosphinooxazoline ligand, (S)-tert-butylPHOX.
- In the synthesis of chiral copper(II) polymers that can catalyze the kinetic resolution of secondary alcohols by acylation.
- In metal-free tandem radical cyclization reactions to synthesize 6-alkyl/acyl phenanthridines.
- In the preparation of tert-leucine-derived N-acetylthiazolidinethione auxiliary that provides high levels of diastereoselection in acetate aldol reactions with a variety of aldehydes.
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![Bicyclo[2.2.1]hepta-2,5-diene-rhodium(I) chloride dimer 96%](/deepweb/assets/sigmaaldrich/product/structures/700/585/b2e5ae1d-2b88-42c8-a071-ef828d4a104c/640/b2e5ae1d-2b88-42c8-a071-ef828d4a104c.png)