699800P
Avanti
MPLA (PHAD™)
Avanti Research™ - A Croda Brand
Synonym(s):
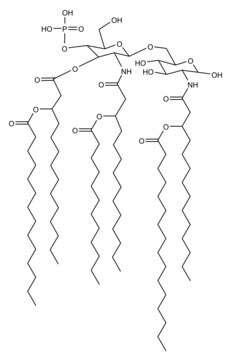
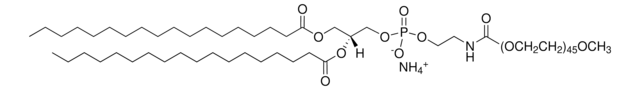
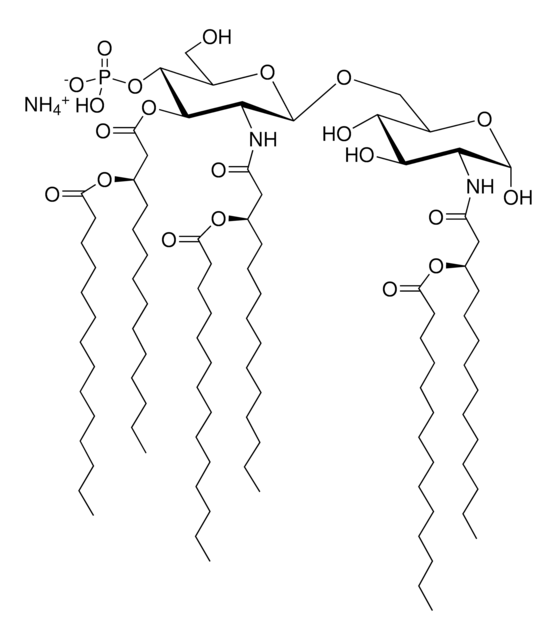
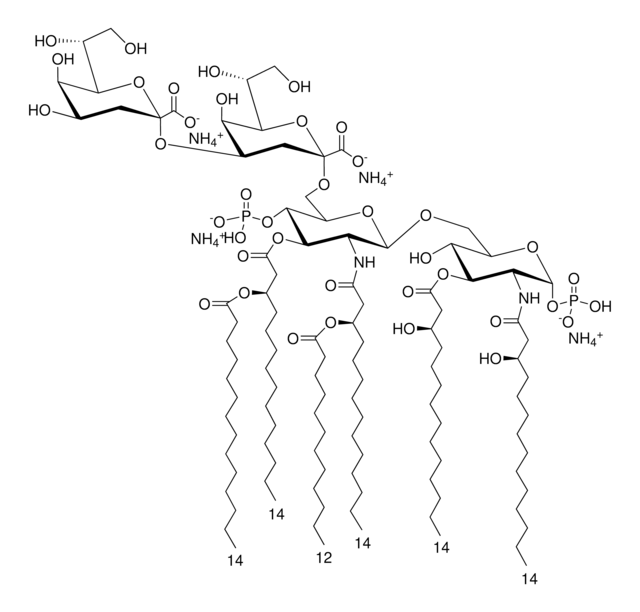
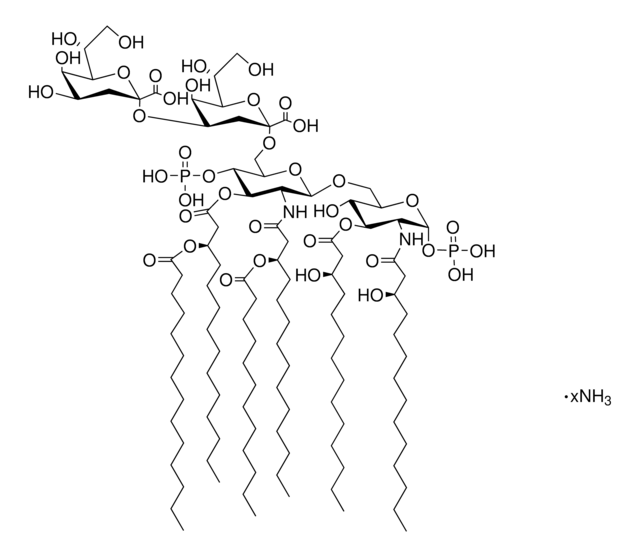
PHAD™ phosphorylated hexaacyl disaccharide; Glycopyranoside Lipid A; GLA
About This Item
Recommended Products
description
Monophosphoryl Lipid A (Synthetic) (PHAD™)
Assay
>99% (HPLC)
form
powder
packaging
pkg of 1 × 1 mg (699800P-1mg)
pkg of 1 × 5 mg (699800P-5mg)
manufacturer/tradename
Avanti Research™ - A Croda Brand
application(s)
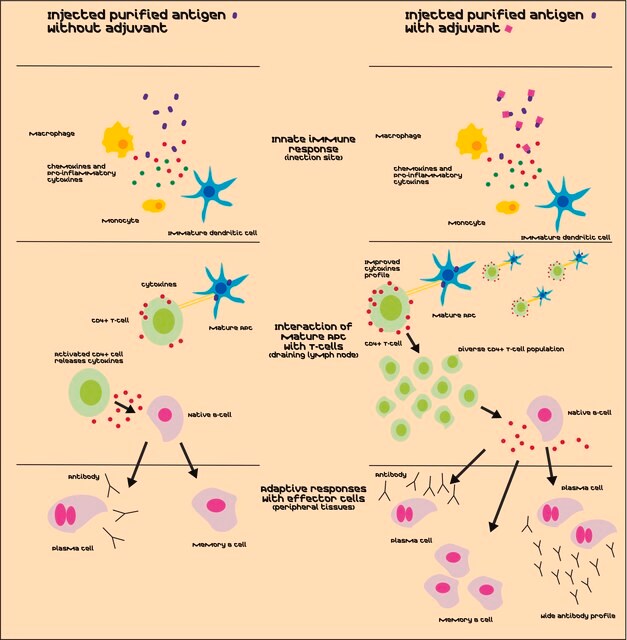
vaccine development
shipped in
dry ice
storage temp.
−20°C
General description
Application
- as a component of cobalt porphyrin-phospholipid (Co-PoP) liposomes for the immunization of mice with membrane proximal external region (MPER) of the gp41 envelope protein
- as an adjuvant along with dimethyldioctadecylammonium bromide(DDA) for C. muridarum recombinant membrane protein based multi-subunit vaccine
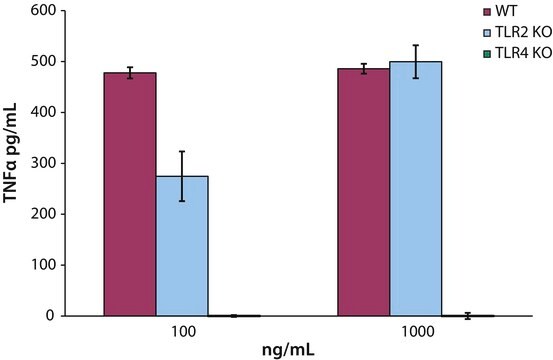
- as toll-like receptor-4 (TLR4) agonist adjuvant for respiratory syncytial virus (RSV) fusion (F) protein FI-RSV vaccine
Biochem/physiol Actions
Packaging
Other Notes
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service