779008
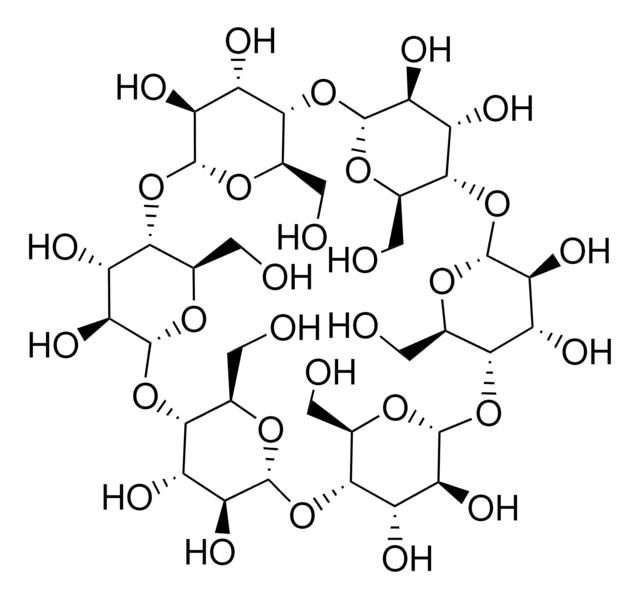
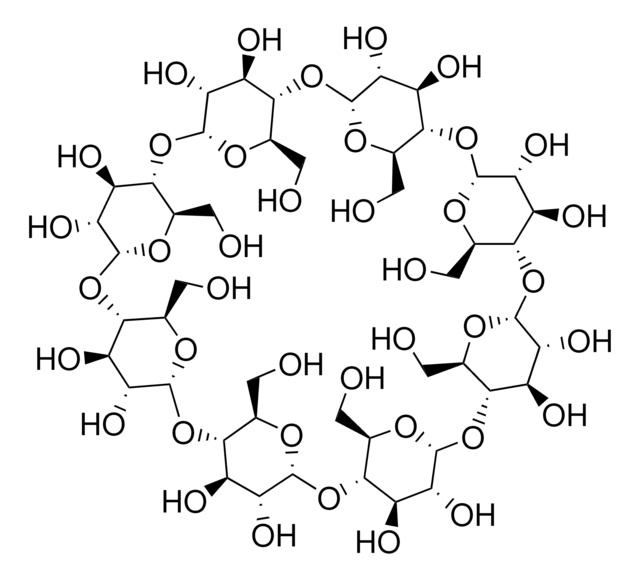
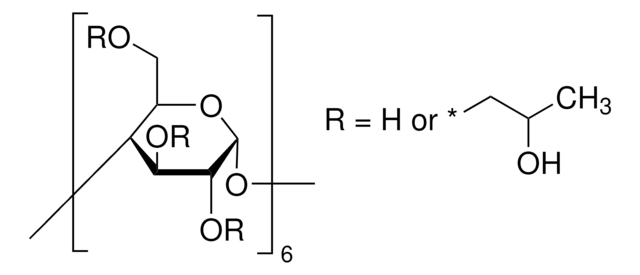
α-Cyclodextrin
Produced by Wacker Chemie AG, Burghausen, Germany, Life Science, Wacker Chemie AG, 98.0-101.0% cyclodextrin basis (HPLC)
Synonym(s):
Cavamax® W6 Pharma, alpha-Cyclodextrin, α-Schardinger dextrin, Cyclohexaamylose, Cyclomaltohexaose
About This Item
Recommended Products
Assay
98.0-101.0% cyclodextrin basis (HPLC)
form
solid
optical activity
[α]/D 147.0 to 152.0° in H2O (USP)
manufacturer/tradename
Wacker Chemie AG
impurities
≤0.20% reducing substances
≤0.25% β- and γ-cyclodextrin (each)
≤0.5% related substances
≤20 ppm residual solvents
≤5 ppm heavy metals (USP)
ign. residue
≤0.10% (USP)
loss
≤10.0% loss on drying
pH
5.0-8.0 (1% in solution)
mp
>278 °C (dec.) (lit.)
absorption
≤0.05 at 350-750 nm in solution at 1%
≤0.10 at 230-250 nm in solution at 1%
suitability
positive for identity (Ph Eur)
functional group
ether
hydroxyl
SMILES string
OC[C@H]1O[C@@H]2O[C@H]3[C@H](O)[C@@H](O)[C@H](O[C@@H]3CO)O[C@H]4[C@H](O)[C@@H](O)[C@H](O[C@@H]4CO)O[C@H]5[C@H](O)[C@@H](O)[C@H](O[C@@H]5CO)O[C@H]6[C@H](O)[C@@H](O)[C@H](O[C@@H]6CO)O[C@H]7[C@H](O)[C@@H](O)[C@H](O[C@@H]7CO)O[C@H]1[C@H](O)[C@H]2O
InChI
1S/C36H60O30/c37-1-7-25-13(43)19(49)31(55-7)62-26-8(2-38)57-33(21(51)15(26)45)64-28-10(4-40)59-35(23(53)17(28)47)66-30-12(6-42)60-36(24(54)18(30)48)65-29-11(5-41)58-34(22(52)16(29)46)63-27-9(3-39)56-32(61-25)20(50)14(27)44/h7-54H,1-6H2/t7-,8-,9-,10-,11-,12-,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23-,24-,25-,26-,27-,28-,29-,30-,31-,32-,33-,34-,35-,36-/m1/s1
InChI key
HFHDHCJBZVLPGP-RWMJIURBSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- As a ligand in the complexation of nonionic surfactants and polyethylene glycols. α-cyclodextrin shows strong interaction with the surfactants that contains no benzene group.
- To protect linoleic acid from oxidation.
- In the synthesis of binuclear copper(II) complexes with cyclodextrins, which can be further used as a template for the preparation of copper nanoparticles incorporated on mesoporous silica.
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service