216690
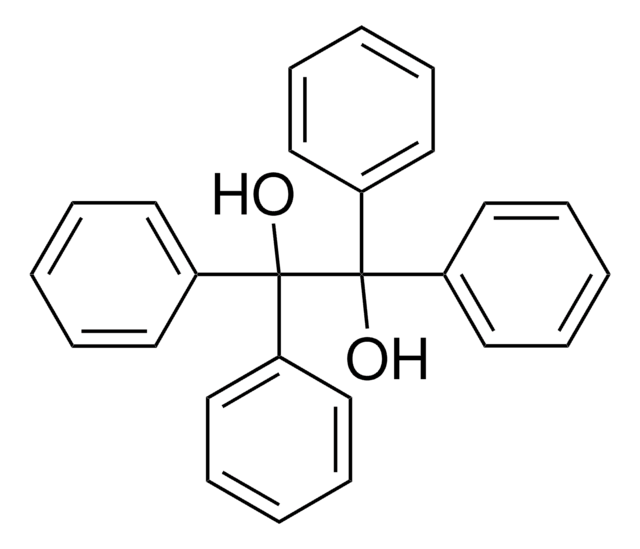
2,2,2-Triphenylacetophenone
98%
Synonym(s):
Benzopinacolone, Phenyl trityl ketone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(C6H5)3CCOC6H5
CAS Number:
Molecular Weight:
348.44
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
mp
182-184 °C (lit.)
functional group
ketone
SMILES string
O=C(c1ccccc1)C(c2ccccc2)(c3ccccc3)c4ccccc4
InChI
1S/C26H20O/c27-25(21-13-5-1-6-14-21)26(22-15-7-2-8-16-22,23-17-9-3-10-18-23)24-19-11-4-12-20-24/h1-20H
InChI key
CFBBKHROQRFCNZ-UHFFFAOYSA-N
General description
Asymmetric reduction of 2,2,2-triphenylacetophenone using with potassium 9-O-(1,2: 5, 6-di-O-isopropylidene-α-D-glucofuranosyl)-9-boratabicyclo [3.3.1] nonane (chiral reducing agent) has been reported. Dehydrative cyclization of 2,2,2-triphenylacetophenone has been reported.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Chiral synthesis via organoboranes. 15. Selective reductions. 42. Asymmetric reduction of representative prochiral ketones with potassium 9-O-(1, 2: 5, 6-di-O-isopropylidene-. alpha.-D-glucofuranosyl)-9-boratabicyclo [3.3. 1] nonane.
Brown HC, etal.
The Journal of Organic Chemistry, 53(6), 1231-1238 (1988)
Douglas A Klumpp et al.
Applied catalysis. A, General, 336(1-2), 128-132 (2008-03-01)
The hydroxyalkylation reactions of aceanthrenequinone (6) and acenapthenequinone (7) with a series of arenes have been studied. In reactions with the Brønsted superacid CF(3)SO(3)H (triflic acid), the condensation products are formed in good yields (58-99%, 10 examples) with high regioselectivity.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service