214973
Diisobutylaluminum hydride solution
1.0 M in methylene chloride
Synonym(s):
DIBAL, DIBAL-H
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
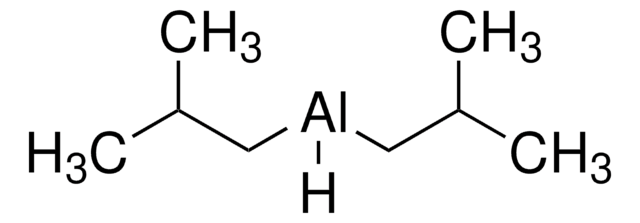
[(CH3)2CHCH2]2AlH
CAS Number:
Molecular Weight:
142.22
Beilstein:
4123663
MDL number:
UNSPSC Code:
12352001
eCl@ss:
38120609
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
liquid
Quality Level
reaction suitability
reagent type: reductant
concentration
1.0 M in methylene chloride
density
1.23 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
CC(C)C[AlH]CC(C)C
InChI
1S/2C4H9.Al.H/c2*1-4(2)3;;/h2*4H,1H2,2-3H3;;
InChI key
AZWXAPCAJCYGIA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Diisobutylaluminum hydride is a reducing agent for many functional groups commonly used in organic synthesis.
Application
Diisobutylaluminum hydride solution (1 M in dichloromethane) has been used in the conversion of esters into α-fluoro-α,β-unsaturated esters in the presence of fluorocarboalkoxy-substituted dialkyl phosphonate anions via Horner-Wadsworth-Emmons reaction. It can be used in the multi-step synthesis of (S)-2-methyl tetrahydropyridine-N-oxide, a key structural moiety of bio-active compounds like himbacine and solenopsin-A.
Used in Pd-catalyzed reductive debromination of secondary alkyl bromides. O-Debenzylation and ring opening of perbenzylated furanosides. Convenient in situ generation of HZrCp2Cl from ZrCp2Cl2 and DIBAL-H.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Carc. 2 - Eye Dam. 1 - Pyr. Liq. 1 - Skin Corr. 1B - STOT SE 3 - Water-react 1
Target Organs
Central nervous system
Supplementary Hazards
Storage Class Code
4.2 - Pyrophoric and self-heating hazardous materials
WGK
WGK 2
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
An enantioselective route to trans-2, 6-disubstituted piperidines
Chackalamannil S
Tetrahedron, 53(32), 11203-11210 (1997)
Reduction-olefination of esters: a new and efficient synthesis of. alpha.-fluoro. alpha.,. beta.-unsaturated esters.
Thenappan A
The Journal of Organic Chemistry, 55(15), 4639-4642 (1990)
Diisobutylaluminum Hydride.
Galatsis P
e-EROS Encyclopedia of Reagents for Organic Synthesis (2008)
Reaction of diisobutylaluminum hydride with selected organic compounds containing representative functional groups
Yoon N M
The Journal of Organic Chemistry, 50(14), 2443-2450 (1985)
Damien Webb et al.
Organic letters, 14(2), 568-571 (2011-12-31)
A continuous flow system for the multiparameter (flow rate, temperature, residence time, stoichiometry) optimization of the DIBALH reduction of esters to aldehydes is described. Incorporating an in-line quench (MeOH), these transformations are generally complete in fewer than 60 s. Mixing
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service