All Photos(1)
About This Item
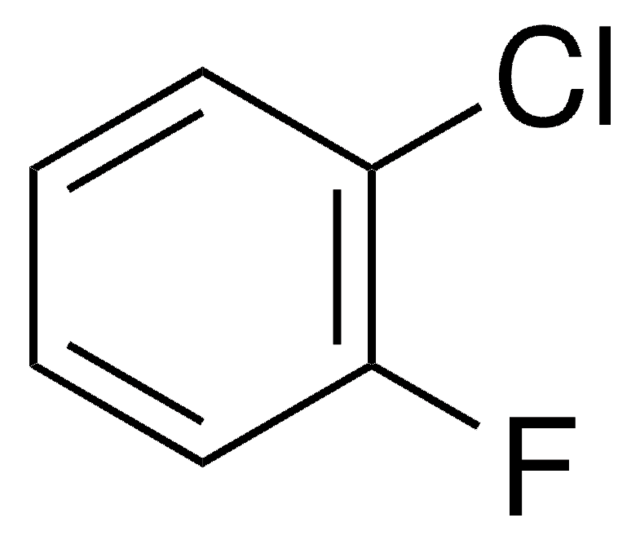
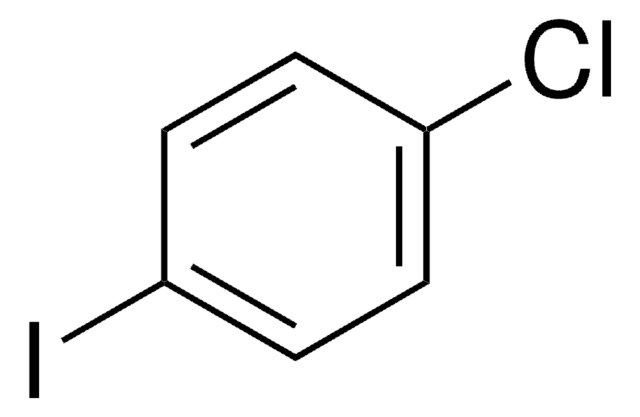
Linear Formula:
ClC6H4F
CAS Number:
Molecular Weight:
130.55
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
liquid
refractive index
n20/D 1.494 (lit.)
bp
126-128 °C (lit.)
density
1.219 g/mL at 25 °C (lit.)
functional group
chloro
fluoro
SMILES string
Fc1cccc(Cl)c1
InChI
1S/C6H4ClF/c7-5-2-1-3-6(8)4-5/h1-4H
InChI key
VZHJIJZEOCBKRA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Reaction of 1-chloro-3-fluorobenzene radical cation with NH3 has been investigated by FT-ICR spectrometry. It reacts with n-butyllithium to yield bicycloadducts via trapping of intermediate benzynes.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
68.0 °F - closed cup
Flash Point(C)
20 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Reactions of dihalobenzene radical cations with ammonia in the gas phase. Reactivity pattern for nucleophilic aromatic substitution.
Tholmann D and Gruetzmacher HF.
Journal of the American Chemical Society, 113(9), 3281-3287 (1991)
Antonio Ramírez et al.
Journal of the American Chemical Society, 126(45), 14700-14701 (2004-11-13)
The key elimination step for the formation of 3-chloro- and 3-fluorobenzyne from 2-chloro-6-fluorophenyllithium displays a pronounced solvent-dependent regioselectivity. 6Li and 13C NMR spectroscopic studies on 2-chloro-6-fluorophenyllithium reveal a single monomeric aryllithium, suggested by DFT computational studies to be a trisolvate.
Oriol Planas et al.
Science (New York, N.Y.), 367(6475), 313-317 (2020-01-18)
Bismuth catalysis has traditionally relied on the Lewis acidic properties of the element in a fixed oxidation state. In this paper, we report a series of bismuth complexes that can undergo oxidative addition, reductive elimination, and transmetallation in a manner
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service