A1153
Aprotinin
3-8 TIU/mg solid, lyophilized powder
Synonym(s):
BPTI, Bovine pancreatic trypsin inhibitor, Trasylol, Trypsin inhibitor (basic)
About This Item
Recommended Products
Product Name
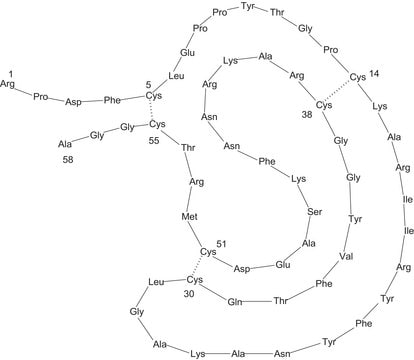
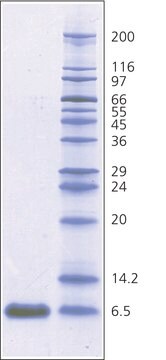
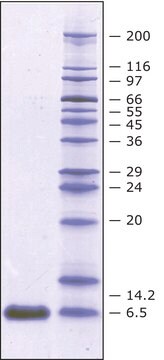
Aprotinin from bovine lung, lyophilized powder, 3-8 TIU/mg solid
biological source
bovine lung
form
lyophilized powder
specific activity
3-8 TIU/mg solid
mol wt
~6,500
solubility
H2O: ≥5 mg/mL
UniProt accession no.
storage temp.
2-8°C
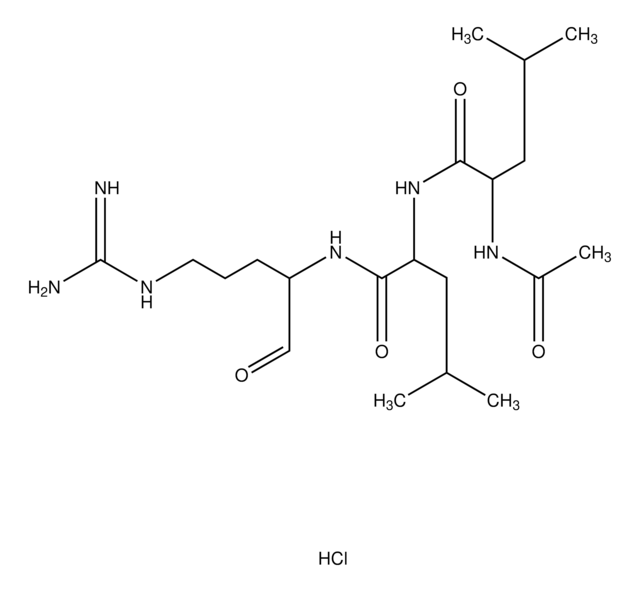
SMILES string
S1SCC2NC(=O)CNC(=O)CNC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C5NC(=O)C(NC(=O)CNC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C(NC(=O)C9N(CCC9)C(=O)CNC(=O)C(NC(=O)C(NC(=O)C%11N(CCC%11)C(=O
InChI
1S/C284H432N84O79S7/c1-21-144(9)222-271(439)337-174(68-46-105-309-282(300)301)239(407)340-187(120-160-77-85-164(374)86-78-160)251(419)341-185(116-156-55-29-24-30-56-156)250(418)342-188(121-161-79-87-165(375)88-80-161)252(420)346-191(123-208(291)378)246(41
InChI key
ZPNFWUPYTFPOJU-UHFFFAOYSA-N
Gene Information
cow ... PTI(404172)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- as a protease inhibitor in radioimmunoprecipitation assay buffer (RIPA) for the homogenization of cardiac microvascular endothelial cells (CMECs)(4) and mammary epithelial cells
- in angiogenesis assay for fibroblast
- in the proteomic stabilization of saliva supernatant
Biochem/physiol Actions
Unit Definition
Preparation Note
also commonly purchased with this product
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
While aprotinin and bovine pancreatic trypsin inhibitor (BPTI) are the same protein sequence, the term aprotinin is typically used when describing the protein derived from bovine lung.
Enzyme Explorer Product Application Index for Elastase. Leukocyte elastase is a 29KDa serine endoprotease of the Proteinase S1 Family. It exists as a single 238 amino acid-peptide chain with four disulfide bonds.
Analytical Enzyme Chymotrypsin: Chymotrypsin is produced in the acinar cells of the pancreas as the inactive precursor, chymotrypsinogen.
Protocols
Objective: To standardize a procedure for the enzymatic assay of Aprotinin.
Related Content
Trypsin is an enzyme in the serine protease class that consists of a polypeptide chain of 223 amino acid residues. Multiple sources, grades and formulations of trypsin specifically designed for research applications are available.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service