P56003
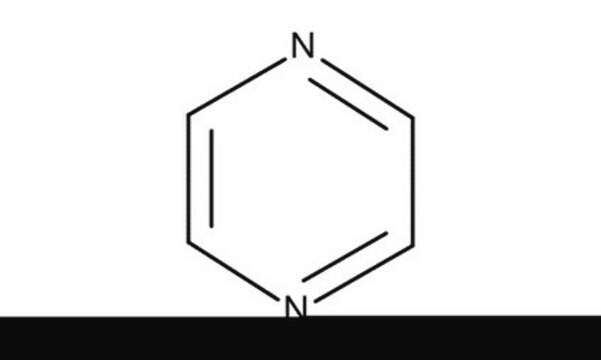
Pyrazine
≥99%
Synonym(s):
1,4-Diazabenzene, 1,4-Diazine, Paradiazine, Piazine, p-Diazine
About This Item
Recommended Products
Quality Level
Assay
≥99%
form
crystals
bp
115-116 °C (lit.)
mp
50-56 °C (lit.)
density
1.031 g/mL at 25 °C (lit.)
SMILES string
c1cnccn1
InChI
1S/C4H4N2/c1-2-6-4-3-5-1/h1-4H
InChI key
KYQCOXFCLRTKLS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- As a bridging ligand in the synthesis of Cu(II) linear chain coordination complexes.
- To prepare Cu(I) derived coordination polymers, which are used in the synthesis of metal-organic frameworks (MOFs).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Sol. 1
Storage Class Code
4.1B - Flammable solid hazardous materials
WGK
WGK 3
Flash Point(F)
131.0 °F - closed cup
Flash Point(C)
55 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
-Cymene; 2,5-Dimethylpyrrole; Acetoin, ≥96%, FCC, FG; 2,5-Dimethylpyrazine; 2,6-Dimethylpyrazine; 2-Ethylpyrazine, ≥98%, FG; 2,3-Dimethylpyrazine; 4-Heptanone; 3-Ethylpyridine; 2,3,5-Trimethylpyrazine; Furfural; Pyrrole; Furfuryl acetate; Linalool; Linalyl acetate; 5-Methylfurfural; γ-Butyrolactone; 2-Acetyl-1-methylpyrrole; Furfuryl alcohol; 2-Acetylpyrrole; Pyrrole-2-carboxaldehyde
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service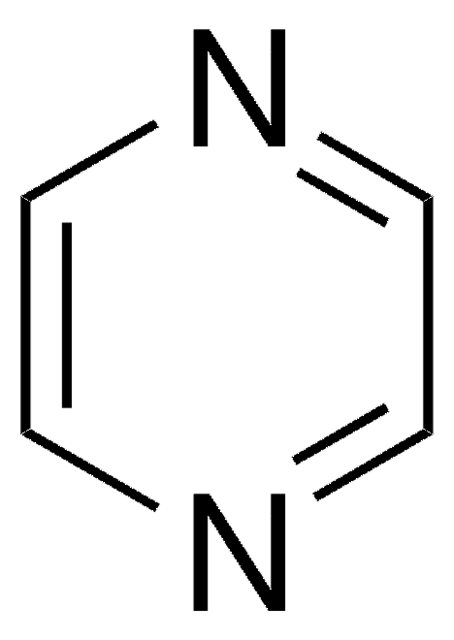

![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)