L2009
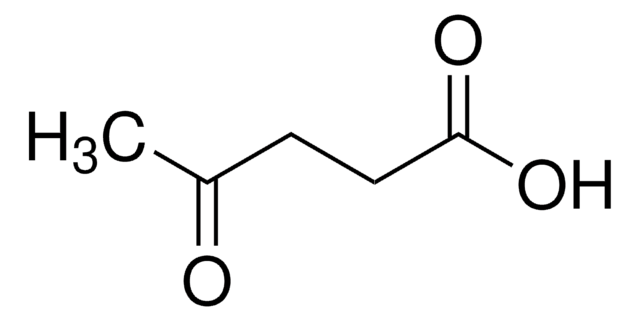
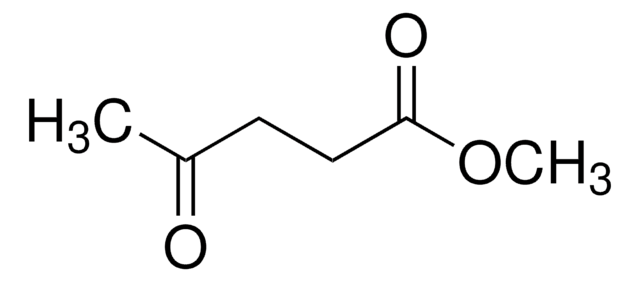
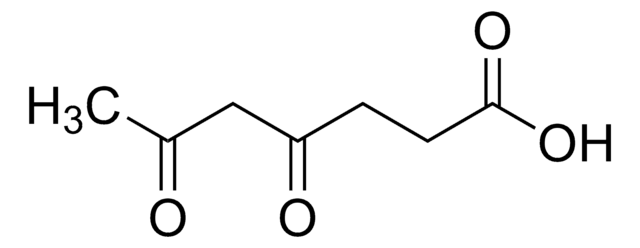
Levulinic acid
98%
Synonym(s):
4-Oxopentanoic acid, 4-Oxovaleric acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3COCH2CH2COOH
CAS Number:
Molecular Weight:
116.12
Beilstein:
506796
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
vapor pressure
1 mmHg ( 102 °C)
Assay
98%
bp
245-246 °C (lit.)
mp
30-33 °C (lit.)
density
1.134 g/mL at 25 °C (lit.)
SMILES string
CC(=O)CCC(O)=O
InChI
1S/C5H8O3/c1-4(6)2-3-5(7)8/h2-3H2,1H3,(H,7,8)
InChI key
JOOXCMJARBKPKM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
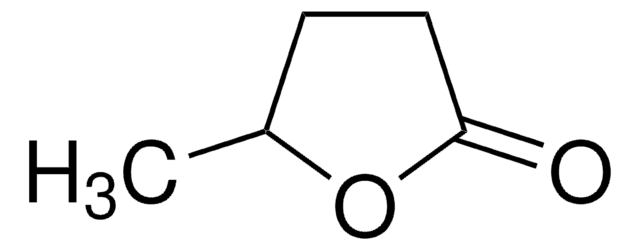
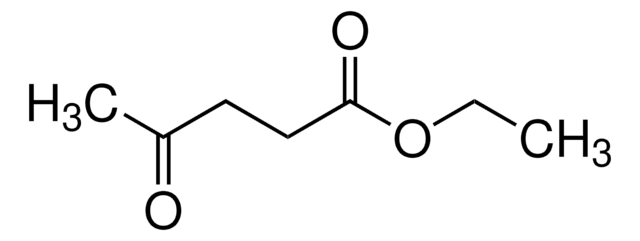
Levulinic acid is a precursor for the synthesis of useful intermediates such as γ-valerolactone, ethyl levulinate, pentanoic acid and 2-methyl-tetrahydrofuran. Derivatization and esterification of levulinic acid results in potential biofuels.
It can also be used in:
It can also be used in:
- The preparation of catalytic composite to synthesize 5-hydroxymethylfurfural and furfural.
- The synthesis of a commercial fragrance, fraistone.
- The synthesis of pyrrolidone derivatives via reductive amination.
- The total synthesis of mycobacterial arabinogalactan.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Sens. 1
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 2
Flash Point(F)
208.4 °F - closed cup
Flash Point(C)
98 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Selective and Efficient Iridium Catalyst for the Reductive Amination of Levulinic Acid into Pyrrolidones.
Wang S, et al.
ChemSusChem, 10(21), 4150-4154 (2017)
Diego Garcés et al.
ChemSusChem, 12(4), 924-934 (2018-12-15)
Homogeneous (HCl) and heterogeneous catalysts (various zeolites) were combined to enhance the upgrading of glucose into two different platform molecules: 5-hydroxymethylfurfural (HMF) and levulinic acid (LA). β-Zeolite was the most active material for the glucose isomerization to fructose, improving also
Vapor Phase Catalytic Transfer Hydrogenation (CTH) of Levulinic Acid to ?-Valerolactone Over Copper Supported Catalysts Using Formic Acid as Hydrogen Source.
Lomate S, et al.
Catalysis Letters, 148(1), 348-358 (2018)
Synthesis of a New Type Perfume Fraistone [J].
Heping L, et al.
Fine Chemicals / ????, 6 (1998)
Nam H Phan et al.
Anti-cancer agents in medicinal chemistry, 18(9), 1349-1355 (2017-11-28)
The aim of this study was to develop and compare polymeric micelles of fucoidan, a sulfated polysaccharide, and hydrophobic drugs such as paclitaxel and curcumin. Paclitaxel and curcumin are both known for their medicinal properties, including anticancer efficacy. However, their
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service