All Photos(2)
About This Item
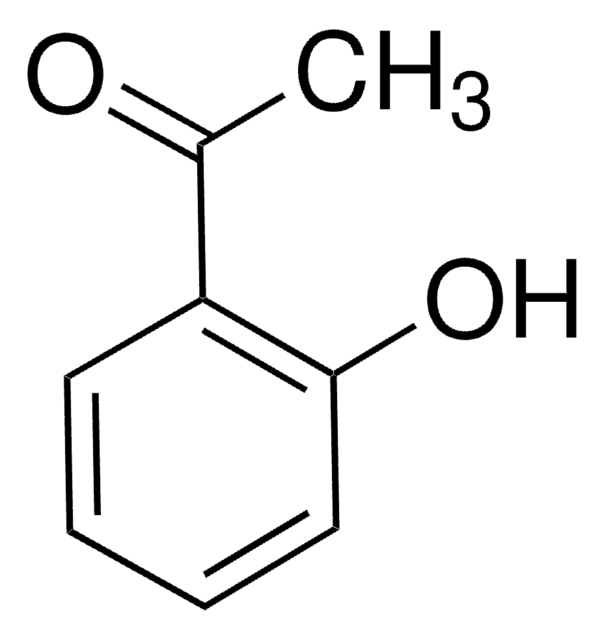
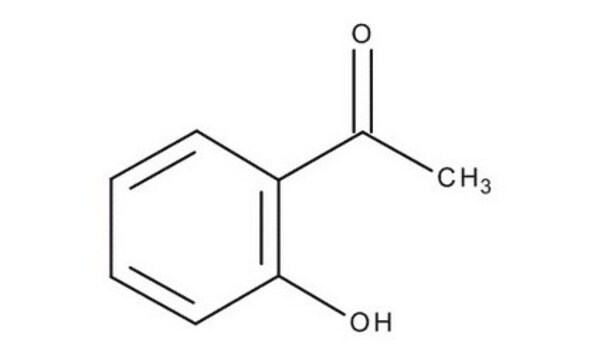
Linear Formula:
C6H5COCH2OH
CAS Number:
Molecular Weight:
136.15
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
86-89 °C (lit.)
functional group
hydroxyl
ketone
phenyl
SMILES string
OCC(=O)c1ccccc1
InChI
1S/C8H8O2/c9-6-8(10)7-4-2-1-3-5-7/h1-5,9H,6H2
InChI key
ZWVHTXAYIKBMEE-UHFFFAOYSA-N
Related Categories
Application
2-Hydroxyacetophenone can be used as a starting material for the synthesis of:
- Enantioselective 1R-phenyl-1,2-ethanediol in the presence of a rhodium(III) catalyst by asymmetric transfer hydrogenation.
- Copper(II) complexes of 2-hydroxyacetophenone N-substituted thiosemicarbazones.
- Chromium, molybdenum, and ruthenium complexes of 2-hydroxyacetophenone Schiff bases.
- 2-Hydroxyacetophenone-aroyl hydrazone derivatives for inhibition of copper corrosion in nitric acid.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Resonance Raman intensity analysis of the excited-state proton transfer in 2-hydroxyacetophenone.
Peteanu LA and Mathies RA.
The Journal of Physical Chemistry, 96(17), 6910-6916 (1992)
F Peng et al.
Letters in applied microbiology, 68(5), 446-454 (2019-02-01)
Chiral 1-phenyl-1,2-ethanediol (PED) performs vital effect for the preparation of pharmaceuticals, agrochemicals and cosmetics. In the study, a newly isolated strain Kurthia gibsoniiSC0312 with the ability to selectively oxidize racemic PED to achieve (S)-PED was evaluated in the aqueous reaction
New copper (II) complexes of 2-hydroxyacetophenone N (4)-substituted thiosemicarbazones and polypyridyl co-ligands: structural, electrochemical and antimicrobial studies
John RP, et al.
Polyhedron, 23(16), 2549-2559 (2004)
Ying-Heng Chen et al.
Journal of agricultural and food chemistry, 65(19), 3965-3974 (2017-04-30)
4-[2-(t-Butylamino)-1-hydroxyethyl]phenol (buctopamine, 4), a new β
Chromium, molybdenum and ruthenium complexes of 2-hydroxyacetophenone Schiff bases
Ali SA, et al.
Journal of Coordination Chemistry, 55(10), 1161-1170 (2002)
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 445134-1KG | |
| 445134-25G | 4061831829891 |
| 445134-5G | 4061831820331 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service