Wichtige Dokumente
B5437
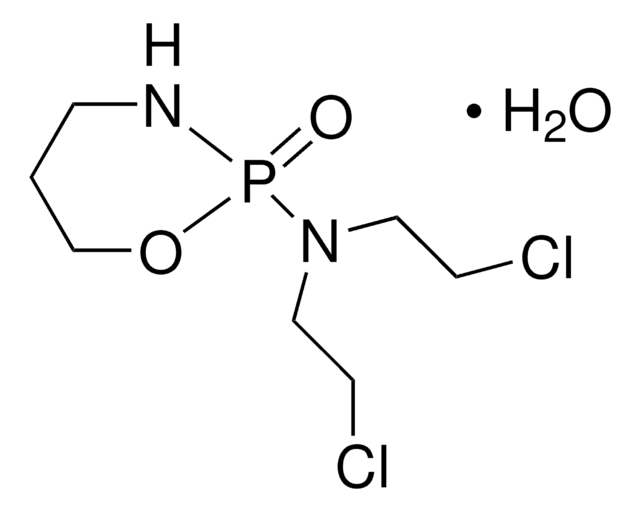
Bendamustine hydrochloride hydrate
≥98% (HPLC)
Synonym(e):
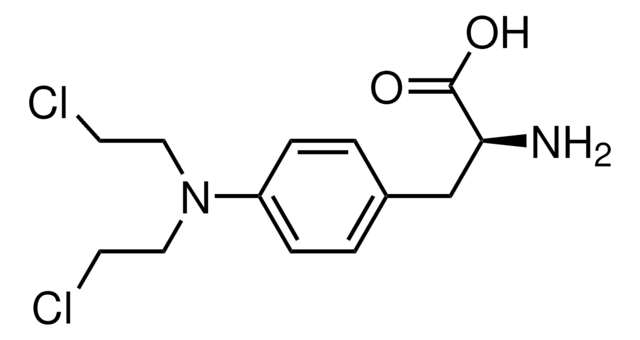
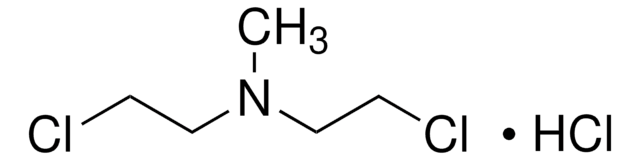
1H-Benzimidazole-2-butanoic acid, 5-[bis(2-chloroethyl)amino]-1-methyl monohydrochloride, Treanda
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥98% (HPLC)
Form
powder
Lagerbedingungen
desiccated
Farbe
off-white
Löslichkeit
H2O: >30 mg/mL
Ersteller
Teva
Lagertemp.
room temp
SMILES String
O.Cl.Cn1c(CCCC(O)=O)nc2cc(ccc12)N(CCCl)CCCl
InChI
1S/C16H21Cl2N3O2.ClH.H2O/c1-20-14-6-5-12(21(9-7-17)10-8-18)11-13(14)19-15(20)3-2-4-16(22)23;;/h5-6,11H,2-4,7-10H2,1H3,(H,22,23);1H;1H2
InChIKey
TWBJYCLUHINEDN-UHFFFAOYSA-N
Allgemeine Beschreibung
Anwendung
- a chemotherapy agent for chronic lymphocytic leukemia (CLL) samples to monitor spliced and unspliced gene expression
- an inhibitor to E3 ubiquitin-protein ligase RNF3 (HOIP) in matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF) assay
- a cytotoxic chemotherapeutic drug in high-throughput screening to test interaction with BAY87-2243
Biochem./physiol. Wirkung
Leistungsmerkmale und Vorteile
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Acute Tox. 3 Oral - Carc. 2 - Muta. 2 - Repr. 2
Lagerklassenschlüssel
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Verwandter Inhalt
n proliferating cells, the cell cycle consists of four phases. Gap 1 (G1) is the interval between mitosis and DNA replication that is characterized by cell growth. Replication of DNA occurs during the synthesis (S) phase, which is followed by a second gap phase (G2) during which growth and preparation for cell division occurs. Together, these three stages comprise the interphase phase of the cell cycle. Interphase is followed by the mitotic (M) phase.
Apoptosis, or programmed cell death (PCD), is a selective process for the removal of unnecessary, infected or transformed cells in various biological systems. As it plays a role in the homeostasis of multicellular organisms, apoptosis is tightly regulated through two principal pathways by a number of regulatory and effector molecules.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.