Alle Fotos(2)
Wichtige Dokumente
A7154
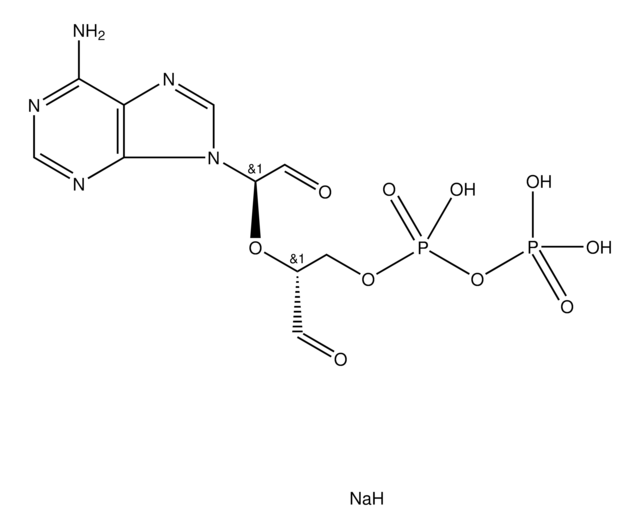
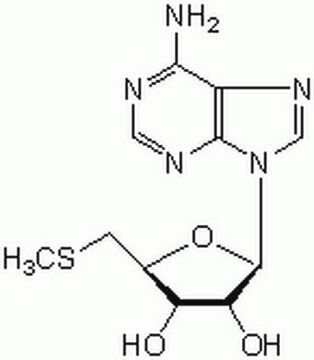
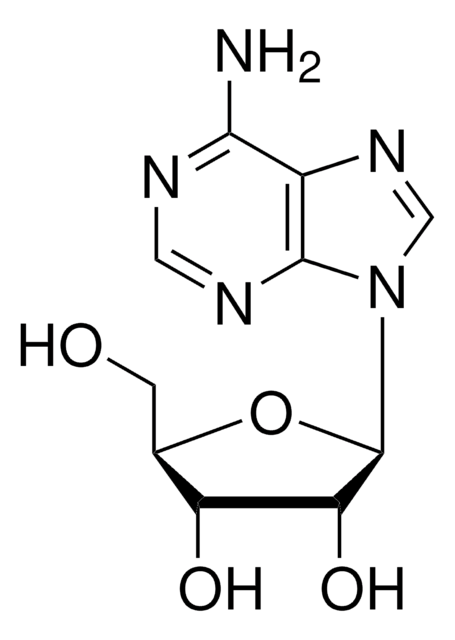
Adenosine, periodate oxidized
≥93%
Synonym(e):
ADOX, Adenosine-2′,3′-dialdehyde
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empirische Formel (Hill-System):
C10H11N5O4
CAS-Nummer:
Molekulargewicht:
265.23
MDL-Nummer:
UNSPSC-Code:
41106305
PubChem Substanz-ID:
NACRES:
NA.51
Empfohlene Produkte
Biologische Quelle
synthetic (organic)
Qualitätsniveau
Assay
≥93%
Form
powder
Löslichkeit
0.2 M HCl: 50 mg/mL, clear, colorless to faintly yellow
Lagertemp.
−20°C
SMILES String
Nc1ncnc2n(cnc12)C(OC(CO)C=O)C=O
InChI
1S/C10H11N5O4/c11-9-8-10(13-4-12-9)15(5-14-8)7(3-18)19-6(1-16)2-17/h1,3-7,17H,2H2,(H2,11,12,13)
InChIKey
ILMNSCQOSGKTNZ-UHFFFAOYSA-N
Anwendung
Adenosine, periodate oxidized has been used:
- as a methylarginine transferase inhibitor in the human embryonic kidney (HEK)-293 T cells
- as a methylase inhibitor in H4 neuroglioma
- as a broad inhibitor of S-adenosylmethionine (AdoMet)-dependent methyltransferases in mouse embryo fibroblast NIH3T3 cells
Biochem./physiol. Wirkung
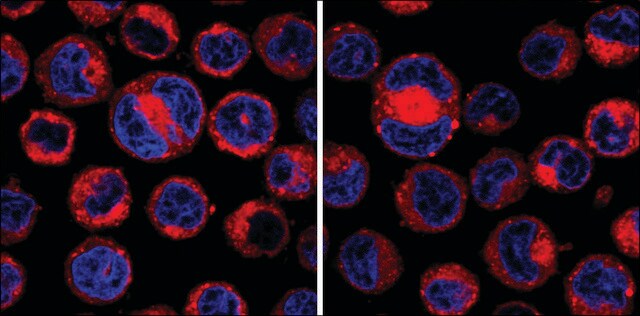
Adenosine, periodate oxidized (Adox) is a protein arginine methyltransferases (PRMTs) inhibitor. It also inhibits the enzyme S-adenosylhomocysteine hydrolase and induces apoptosis. Its inhibitory effect on histone methyltransferases prevents histone methylation. Adox also elicits intrinsic cytotoxic properties.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Ryan Heit et al.
Journal of cell science, 122(Pt 16), 2957-2968 (2009-07-30)
Trimethylation of lysine 9 on histone H3 (H3K9me3) is known both to be necessary for proper chromosome segregation and to increase in late G2. We investigated the role of late G2 methylation, specifically in mitotic progression, by inhibiting methylation for
Christian Schwerk et al.
Oncogene, 24(47), 7002-7011 (2005-07-12)
We have analysed the importance of proper substrate methylation by S-adenosylmethionine-dependent methyltransferases for cell survival and cell cycle progression. We show that treatment of cells with the methyltransferase inhibitor adenosine dialdehyde (AdOx) causes cell cycle arrest and death in different
Olga Blifernez et al.
The Plant journal : for cell and molecular biology, 65(1), 119-130 (2010-12-24)
Methylation of protein arginines represents an important post-translational modification mechanism, which has so far primarily been characterized in mammalian cells. In this work, we successfully identified and characterized arginine methylation as a crucial type of post-translational modification in the activity
Björn Hultberg
Clinica chimica acta; international journal of clinical chemistry, 356(1-2), 117-124 (2005-05-05)
Many clinical and epidemiological studies show that mild hyperhomocysteinemia is associated with premature vascular disease. Information about the metabolism of homocysteine is therefore essential for an understanding of its role in atherogenesis, thereby enabling a modulation of that risk. In
Yinghong He et al.
Journal of translational medicine, 11, 14-14 (2013-01-16)
Pharmacologic reactivation of fetal hemoglobin expression is a promising strategy for treatment of sickle cell disease and β-thalassemia. The objective of this study was to investigate the effect of the methyl transferase inhibitor adenosine-2',3'-dialdehyde (Adox) on induction of human fetal
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.