Y0000155
Tramadolhydrochlorid -hydrochlorid
European Pharmacopoeia (EP) Reference Standard
Synonym(e):
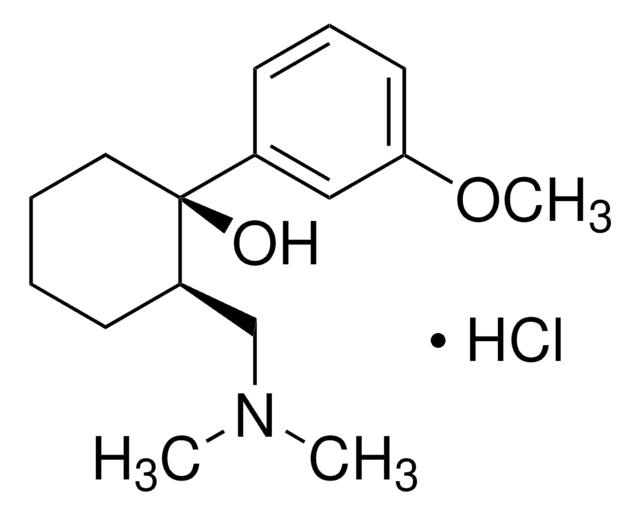
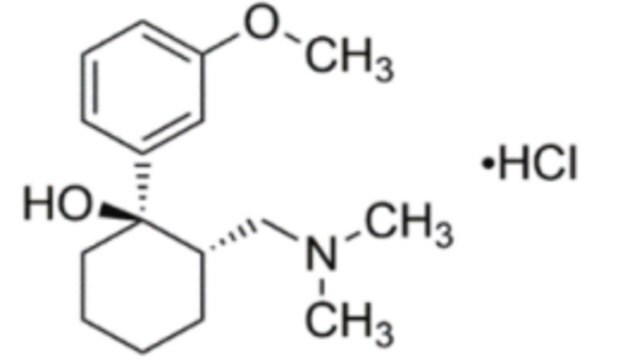
(±)-cis-2-(Dimethylaminomethyl)-1-(3-methoxyphenyl)-cyclohexanol -hydrochlorid -hydrochlorid
About This Item
Empfohlene Produkte
Qualität
pharmaceutical primary standard
API-Familie
tramadol
Hersteller/Markenname
EDQM
Arzneimittelkontrolle
USDEA Schedule IV
Anwendung(en)
pharmaceutical (small molecule)
Format
neat
Lagertemp.
2-8°C
SMILES String
Cl.COc1cccc(c1)[C@@]2(O)CCCC[C@@H]2CN(C)C
InChI
1S/C16H25NO2.ClH/c1-17(2)12-14-7-4-5-10-16(14,18)13-8-6-9-15(11-13)19-3;/h6,8-9,11,14,18H,4-5,7,10,12H2,1-3H3;1H/t14-,16+;/m1./s1
InChIKey
PPKXEPBICJTCRU-XMZRARIVSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Tramadol is a synthetic opiate agonist analgesic, widely used in patients suffering from moderate to severe chronic pain. It is considered safe when compared to other opioids. It contributes to the analgesic activity by blocking the nociceptive impulses at the spinal level, inhibiting the norepinephrine and serotonin reuptake.
Anwendung
Biochem./physiol. Wirkung
Verpackung
Sonstige Hinweise
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Acute Tox. 3 Oral - Aquatic Chronic 2 - STOT SE 3
Zielorgane
Central nervous system
Lagerklassenschlüssel
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 2
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Leider sind derzeit keine COAs für dieses Produkt online verfügbar.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.