PHR1018
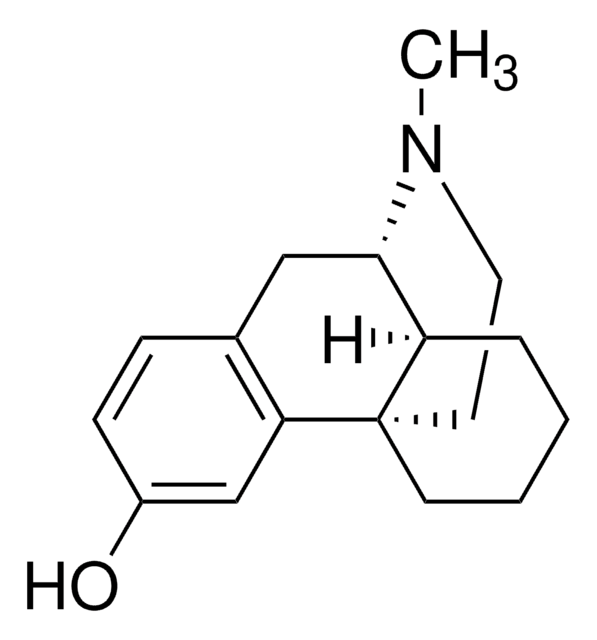
Dextromethorphan HBr
certified reference material, pharmaceutical secondary standard
Synonym(e):
Dextromethorphan -hydrobromid Monohydrat, (9S,13S,14S)-3-Methoxy-17-methylmorphinan -hydrobromid
About This Item
Qualität
certified reference material
pharmaceutical secondary standard
Qualitätsniveau
Agentur
traceable to Ph. Eur. D0740000
traceable to USP 1181007
API-Familie
dextromethorphan
Analysenzertifikat (CofA)
current certificate can be downloaded
Methode(n)
HPLC: suitable
gas chromatography (GC): suitable
Anwendung(en)
pharmaceutical (small molecule)
Format
neat
Lagertemp.
2-30°C
SMILES String
Br[H].[H]O[H].[H][C@]12CCCC[C@]13CCN(C)[C@H]2Cc4ccc(OC)cc34
InChI
1S/C18H25NO.BrH.H2O/c1-19-10-9-18-8-4-3-5-15(18)17(19)11-13-6-7-14(20-2)12-16(13)18;;/h6-7,12,15,17H,3-5,8-11H2,1-2H3;1H;1H2/t15-,17+,18+;;/m1../s1
InChIKey
STTADZBLEUMJRG-IKNOHUQMSA-N
Angaben zum Gen
human ... GRIN2A(2903) , SIGMAR1(10280)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Anwendung
Hinweis zur Analyse
Sonstige Hinweise
Fußnote
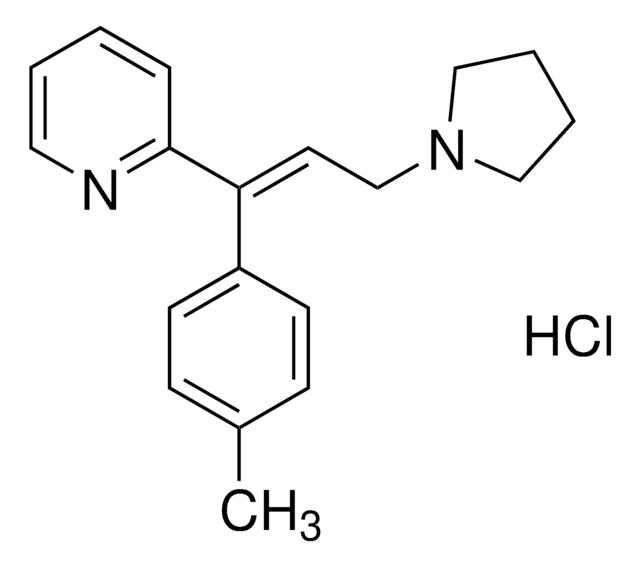
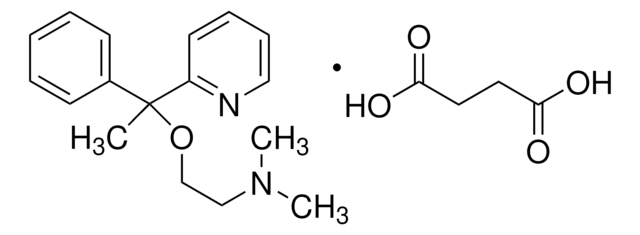
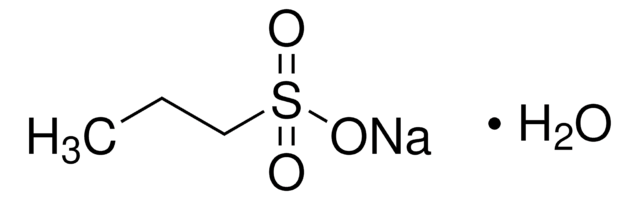
Ähnliches Produkt
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Acute Tox. 3 Oral - Aquatic Chronic 2
Lagerklassenschlüssel
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
WGK
WGK 2
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Choose from one of the most recent versions:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Protokolle
Silica gel G 254 plates are suitable for analysis of Dextromethorphan following the European pharmacopeia monograph.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.