Alle Fotos(3)
Wichtige Dokumente
523976
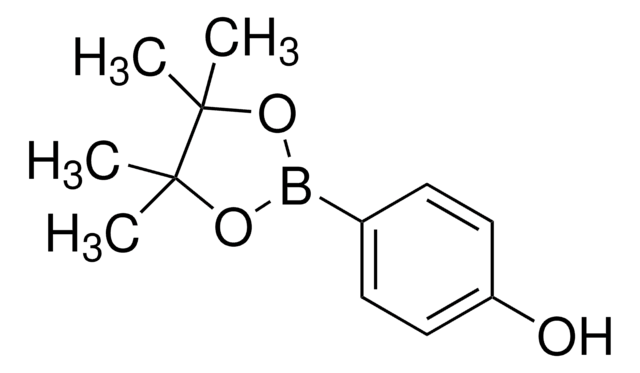
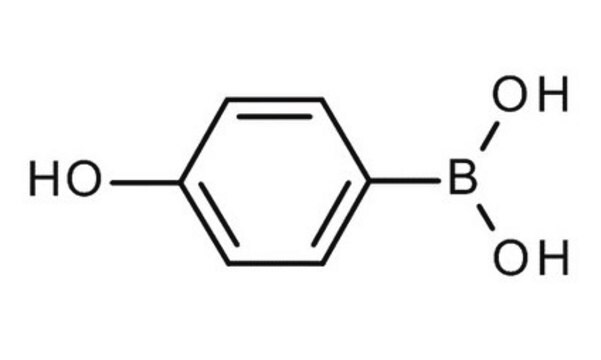
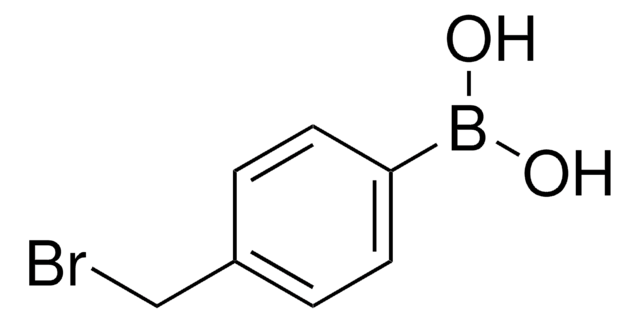
4-Hydroxyphenylborsäure
≥95.0%
Synonym(e):
(p-Hydroxyphenyl)boronic acid, 4-Hydroxybenzeneboronic acid, p-hydroxy-benzeneboronic acid
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(3)
About This Item
Lineare Formel:
HOC6H4B(OH)2
CAS-Nummer:
Molekulargewicht:
137.93
MDL-Nummer:
UNSPSC-Code:
12352103
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
≥95.0%
Form
solid
mp (Schmelzpunkt)
>230 °C (lit.)
SMILES String
OB(O)c1ccc(O)cc1
InChI
1S/C6H7BO3/c8-6-3-1-5(2-4-6)7(9)10/h1-4,8-10H
InChIKey
COIQUVGFTILYGA-UHFFFAOYSA-N
Verwandte Kategorien
Anwendung
4-Hydroxyphenylboronic acid can be used as a reactant in:
It can also be used to prepare/promote:
- Suzuki-Miyaura coupling and Stille coupling reactions.
- Palladium-catalyzed aminocarbonylation and cross-coupling reactions.
- Suzuki reaction for preparation of bio-supported palladium nanoparticles as phosphine-free catalysts.
- Cu2O-catalyzed aerobic oxidative cross-coupling of tetrazoles.
It can also be used to prepare/promote:
- PDK1 inhibitory activity (cancer cell growth, survival, and tumorigenesis inhibitor).
- Rod-like dendronized polymers containing G4 and G5 ester dendrons via macromonomer approach by living ROMP.
- Estrone-derived cyclopamine analogs as Sonic Hedgehog signaling inhibitors for anti-cancer chemotherapeutics.
- Enzymatic inhibitors for the treatment of Gram-negative bacterial infections.
- Oligoarenes by Suzuki-Miyaura palladium-catalyzed cross-coupling.
Sonstige Hinweise
Contains varying amounts of anhydride
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Zhaoyang Lu et al.
Soft matter, 12(17), 3860-3867 (2016-03-31)
The self-assembling behavior of coil-rod-coil molecules 1a, 1b, and 2a, 2b was investigated using DSC, POM, SAXS, and AFM in bulk and aqueous solutions. These molecules contain p-quinquephenyl groups as rod segments incorporating lateral hydroxyl or methoxyl groups in the
Stéphanie Blanchard et al.
Bioorganic & medicinal chemistry letters, 22(8), 2880-2884 (2012-03-23)
A series of 2-anilino substituted 4-aryl-8H-purines were prepared as potent inhibitors of PDK1, a serine-threonine kinase thought to play a role in the PI3K/Akt signaling pathway, a key mediator of cancer cell growth, survival and tumorigenesis. The synthesis, SAR and
Synthetic approach to the chemical isostere of O-methyl honokiol
Cui, M.; Kim, H. S.
Synlett, 23, 311-313 (2012)
Highly selective palladium-catalyzed aminocarbonylation and cross-coupling reactions on a cavitand scaffold
Csok, Z.; Takatsy, A.; Kollar, L.
Tetrahedron, 68, 2657-2661 (2012)
Synthesis of Rod-Like Dendronized Polymers Containing G4 and G5 Ester Dendrons via Macromonomer Approach by Living ROMP
Kim, K.O.; Choi, T-L.
ACS Macro Letters, 1, 445-448 (2012)
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 523976-5G | 4061832549460 |
| 523976-1G | 4061832549354 |
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.