Alle Fotos(1)
Wichtige Dokumente
436860
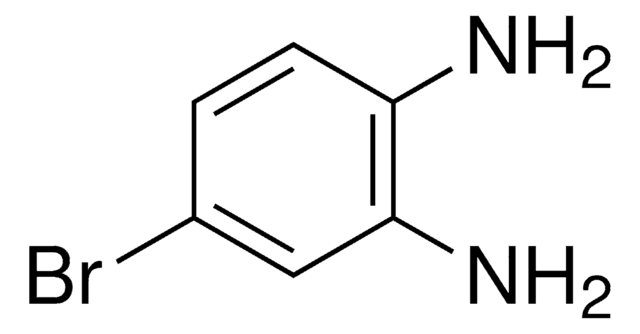
N,N-Diisopropylanilin
97%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
C6H5N[CH(CH3)2]2
CAS-Nummer:
Molekulargewicht:
177.29
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
97%
Form
liquid
Brechungsindex
n20/D 1.519 (lit.)
bp
95-96 °C/11 mmHg (lit.)
Dichte
0.91 g/mL at 25 °C (lit.)
Funktionelle Gruppe
amine
SMILES String
CC(C)N(C(C)C)c1ccccc1
InChI
1S/C12H19N/c1-10(2)13(11(3)4)12-8-6-5-7-9-12/h5-11H,1-4H3
InChIKey
OVSARSKQWCLSJT-UHFFFAOYSA-N
Allgemeine Beschreibung
N,N-Diisopropylaniline is an N,N-dialkylaniline. Its borane adducts have been prepared and reported as hydroborating agents. Thermal dissociation of the borane-N,N-diisopropylaniline adduct has been reported to afford gaseous diborane. N,N-Diisopropylaniline is a diisoproplyamino derivative that can be prepared by reacting bromobenzene with diisopropylamine.
Anwendung
N,N-Diisopropylaniline was used for the synthesis of 4-diisopropylamino benzonitrile. It may be used for the synthesis of 6-(4-bromophenyl)-3-methoxy-5-methyl-8-oxabicyclo[3.2.1]octa-3,6-dien-2-one and N,N,N′,N′-tetraisopropylbenzidine.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
10 - Combustible liquids
WGK
WGK 3
Flammpunkt (°F)
183.2 °F - closed cup
Flammpunkt (°C)
84 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Yvonne D Williams et al.
The Journal of organic chemistry, 78(23), 11707-11713 (2013-11-01)
Methoxytropolones are useful scaffolds for therapeutic development because of their known biological activity and established value in the synthesis of α-hydroxytropolones. Upon treatment with triflic acid, a series of 3-methoxy-8-oxabicyclo[3.2.1]octa-3,6-dien-2-ones rearrange rapidly and cleanly to form methoxytropolones. Interestingly, bicycles that
N-alkylation of hindered secondary aromatic amines with 2-iodobutane.
Katritzky AR, et al.
ORL; Journal for Oto-Rhino-Laryngology and Its Related Specialties, 23(4), 399-402 (1991)
Synthesis of N,N,N',N'-tetraalkylbenzidines through oxidative coupling of N,N-dialkylarylamines induced by SbCl5.
Vitale P, et al.
ARKIVOC (Gainesville, FL, United States), 3, 36-48 (2013)
Dual fluorescence and fast intramolecular charge transfer with 4-(diisopropylamino) benzonitrile in alkane solvents.
Demeter A, et al.
Chemical Physics Letters, 323(3), 351-360 (2000)
Brown et al.
The Journal of organic chemistry, 65(15), 4655-4661 (2000-08-26)
Several N,N-diethyl-tert-alkylamines, such as N,N-diethyl-2-methyl-2-butylamine (1, t-PentNEt2), N,N-diethyl-2,3-dimethyl-2-butylamine (2, t-HexNEt2), N,N-diethyl-2,3,3-trimethyl-2-butylamine (3, t-HeptNEt2), and N,N-diethyl-1,1,3,3-tetramethylbutylamine (4, t-OctNEt2) with varying steric bulk around nitrogen (by changing the tert-alkyl group) have been prepared and examined as borane carriers. The complexing ability of
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.