408085
3-Brom-5-iodbenzoesäure
97%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
BrC6H3(I)CO2H
CAS-Nummer:
Molekulargewicht:
326.91
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
97%
Form
solid
mp (Schmelzpunkt)
219-221 °C (lit.)
Funktionelle Gruppe
bromo
carboxylic acid
iodo
SMILES String
OC(=O)c1cc(Br)cc(I)c1
InChI
1S/C7H4BrIO2/c8-5-1-4(7(10)11)2-6(9)3-5/h1-3H,(H,10,11)
InChIKey
MKJBJYCBKXPQSY-UHFFFAOYSA-N
Allgemeine Beschreibung
3-Bromo-5-iodobenzoic acid (BrIBA) is a halogen substituted carboxylic acid.
Anwendung
3-Bromo-5-iodobenzoic acid may be used in the preparation of the following:
- Phenyl(3-bromo-5-iodo)benzoate.
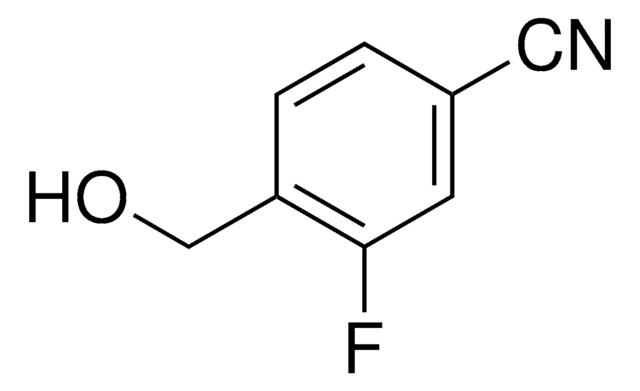
- As starting reagent for the large-scale synthesis of the thromboxane receptor antagonist 3-{3-[2-(4-chlorobenzenesulfonamido)ethyl]-5-(4-fluorobenzyl)phenyl}propionic acid, via regioselective Heck cross-coupling reaction.
- Methyl 3-bromo-5-iodobenzoate.
- 3-Bromo-5-(triisopropylsilylethynyl)benzoic acid, via Sonogashira coupling reaction.
- Trifluoroacetophenone.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Synthesis of acetylene-functionalized [2]rotaxane monomers directed toward side chain-type polyrotaxanes.
Nakazono K, et al.
Polymer Journal, 42(3), 208-215 (2010)
A scalable synthesis of the thromboxane receptor antagonist 3-{3-[2-(4-chlorobenzenesulfonamido) ethyl]-5-(4-fluorobenzyl) phenyl} propionic acid via a regioselective Heck cross-coupling strategy.
Waite DC and Mason CP.
Organic Process Research & Development, 2(2), 116-120 (1998)
S Lindman et al.
Bioorganic & medicinal chemistry, 8(9), 2375-2383 (2000-10-12)
Rigidification of peptides by cyclization and iterative incorporation of well-defined secondary structure mimetics constitutes one approach to the design of non-peptidergic structures with better defined conformations. We herein present the synthesis of a potential gamma-turn mimetic scaffold, and its incorporation
Keisuke Gondo et al.
Molecules (Basel, Switzerland), 17(6), 6625-6632 (2012-06-26)
Reaction of dibenzoylmethane with (diacetoxyiodo)benzene in the presence of KOH in MeCN quantitatively gave the corresponding iodonium ylide, which was treated with a HF reagent to afford the corresponding 2-fluorinated dibenzoylmethane in 14-50% yields. The similar reaction of the iodonium
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.
![3-[4-(Dimethylamino)phenyl]propanoic acid hydrochloride AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/161/863/4d4f0008-ec42-42c7-9f1f-c03aec8be0e0/640/4d4f0008-ec42-42c7-9f1f-c03aec8be0e0.png)