Alle Fotos(1)
Wichtige Dokumente
383503
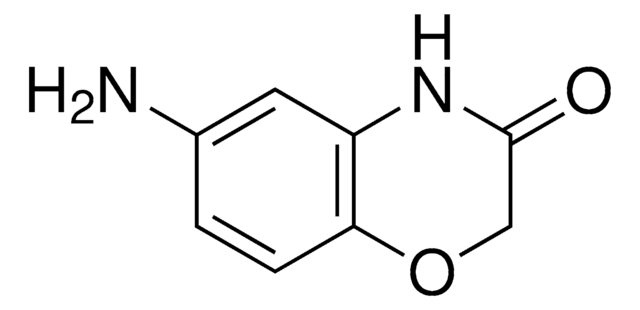
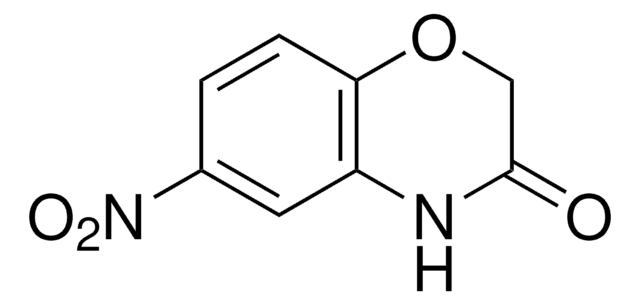
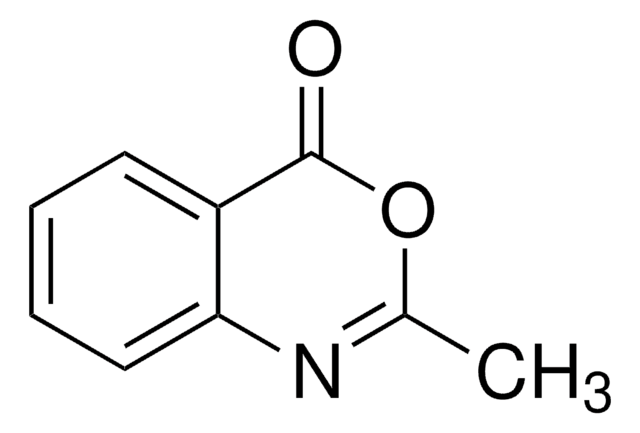
2H-1,4-Benzoxazin-3(4H)-on
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C8H7NO2
CAS-Nummer:
Molekulargewicht:
149.15
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
99%
mp (Schmelzpunkt)
173-175 °C (lit.)
Löslichkeit
methanol: soluble 25 mg/mL, clear, colorless
SMILES String
O=C1COc2ccccc2N1
InChI
1S/C8H7NO2/c10-8-5-11-7-4-2-1-3-6(7)9-8/h1-4H,5H2,(H,9,10)
InChIKey
QRCGFTXRXYMJOS-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
2H-1,4-Benzoxazin-3(4H)-one, a benzoxazine derivative, is a heterocyclic building block for various natural and synthetic organic compounds. It has been reported as an intermediate during the biogenesis of cyclic hydoxamic acids in maize. Its standard molar enthalpy of formation and tautomerization energy of its tautomers has been evaluated by calorimetric and computational methods. It has been synthesized by reacting o-aminophenol with chloroacetyl chloride in the presence of butanone and aqueous NaHCO3.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
2H-1,4-benzoxazin-3(4H)-one, an intermediate in the biosynthesis of cyclic hydroxamic acids in maize.
Kumar P, et al.
Phytochemistry, 36(4), 893-898 (1994)
A general and convenient synthesis of 2H-1,4-benzoxazin-3(4H)-ones.
Shridhar DR, et al.
Organic Prep. and Proc. Int., 14(3), 195-197 (1982)
Jiu Hong Wu et al.
Bioorganic & medicinal chemistry letters, 13(13), 2223-2225 (2003-06-12)
A new inhibitor of in vitro tumor cell replication, cappamensin A (1) (2H-1,4-benzoxazin-3(4H)-one, 6-methoxy-2-methyl-4-carbaldehyde), was isolated from the roots of Capparis sikkimensis subsp. formosana using bioactivity-guided fractionation. The structure of 1 was established by spectroscopic methods, including 2D NMR analyses.
Calorimetric and computational study of 2H-1,4-benzoxazin-3(4H)-one and of related species.
Matos MAR, et al.
Molecular Physics, 104(12), 1833-1841 (2006)
Recent advances in the synthesis of 2H-1, 4-benzoxazin-3-(4H)-ones and 3, 4-dihydro-2 H -1, 4-benzoxazines.
Ilas J, et al.
Tetrahedron, 61(31), 7325-7348 (2005)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.