SMB00445
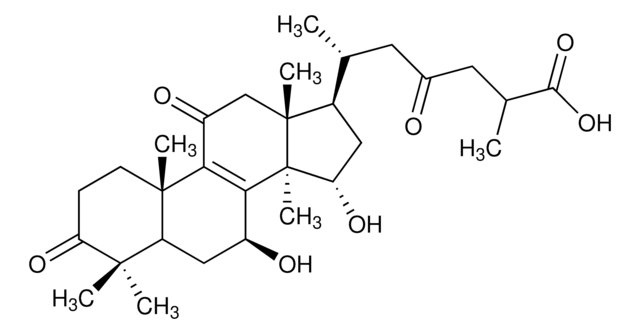
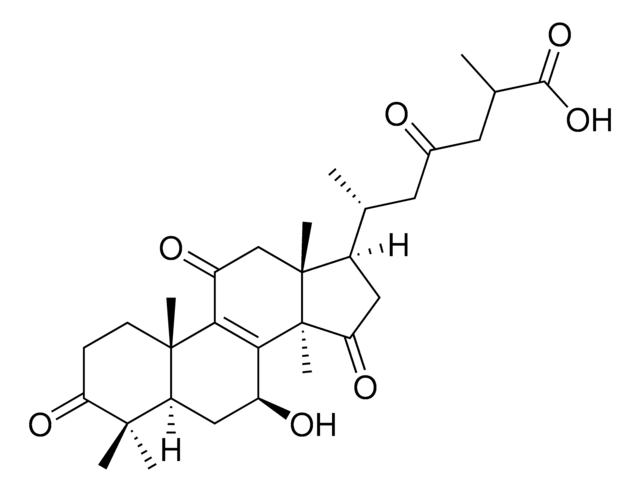
Ganoderic acid A
≥98% (HPLC)
Synonym(s):
(2R,6R)-6-[(5R,7S,10S,13R,14R,15S,17R)-7,15-dihydroxy-4,4,10,13,14-pentamethyl-3,11-dioxo-2,5,6,7,12,15,16,17-octahydro-1H-cyclopenta[a]phenanthren-17-yl]-2-methyl-4-oxoheptanoic acid, (7β,15α,25R)-7,15-Dihydroxy-3,11,23-trioxo-lanost-8-en-26-oic acid
About This Item
Recommended Products
Quality Level
Assay
≥98% (HPLC)
form
powder
application(s)
metabolomics
vitamins, nutraceuticals, and natural products
storage temp.
−20°C
SMILES string
OC(C(C)CC(C[C@@H](C)[C@H]1C[C@H](O)[C@@]([C@]1(C)CC2=O)(C)C3=C2[C@]4(C)C(C[C@@H]3O)C(C)(C)C(CC4)=O)=O)=O
InChI
1S/C30H44O7/c1-15(10-17(31)11-16(2)26(36)37)18-12-23(35)30(7)25-19(32)13-21-27(3,4)22(34)8-9-28(21,5)24(25)20(33)14-29(18,30)6/h15-16,18-19,21,23,32,35H,8-14H2,1-7H3,(H,36,37)/t15-,16?,18-,19+,21?,23+,28+,29-,30+/m1/s1
InChI key
DYOKDAQBNHPJFD-ZQEHRSJRSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- to study its protective effects on hypoxia-induced rat cardiomyocytes (H9c2) cell injury
- as a reference standard to study its inhibitory and antiviral effects against groundnut bud necrosis virus (GBNV) infection in cowpea plants
- as a standard in Fourier transformed-infrared (FT-IR) spectroscopy for the analysis of secondary metabolites from Ganoderma lucidum
Biochem/physiol Actions
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service